Volume 5 (2022) Issue 3 No.3 Pages 79-82
Abstract
We report the case of a 28-year-old woman who developed upper abdominal pain and jaundice after a second unrelated allogeneic hematopoietic cell transplantation (allo-HCT) for acute lymphoid leukemia (ALL). Laboratory data showed elevated levels of liver enzymes, amylase, and lipase. Although acute pancreatitis was suspected, no structural lesions were detected. Liver biopsy was compatible with late-onset acute graft-versus-host disease (GVHD), which resolved following treatment with methylprednisolone (mPSL) and tacrolimus (TAC). In addition, her serum amylase level and abdominal pain rapidly resolved following acute GVHD-directed therapy. Acute pancreatitis concomitant with late-onset acute liver GVHD is extremely rare and has not been documented subsequent to a second allo-HCT.
Introduction
Acute graft-versus-host disease (aGVHD) is a major complication of allogeneic hematopoietic cell transplantation (allo-HCT). aGVHD occurs when donor T cells recognize the host cells as
Case Report
A 28-year-old woman with a history of ALL was admitted to our hospital for upper abdominal pain. She was diagnosed with ALL in 2016 and achieved complete remission (CR) following standard chemotherapy. She then underwent her first allogeneic bone marrow transplantation (allo-BMT) from a human leukocyte antigen (HLA)-matched unrelated donor in October 2016 but relapsed in 2018. A second CR was achieved using multidrug chemotherapy. As consolidative therapy, she underwent a second allo-HCT from a matched unrelated peripheral blood stem cell donor in November 2018. Conditioning consisted of fludarabine, melphalan, and 4 Gy of total body irradiation. GVHD prophylaxis included TAC and short-term methotrexate administration. Bone marrow evaluation on day 28 showed complete hematological remission and full donor chimerism. Because the patient had no aGVHD, TAC was slowly tapered off. However, late-onset liver aGVHD developed (stage 1, overall grade II) on day 119; therefore, prednisolone was started at a dose of 20 mg/day, and TAC was resumed.
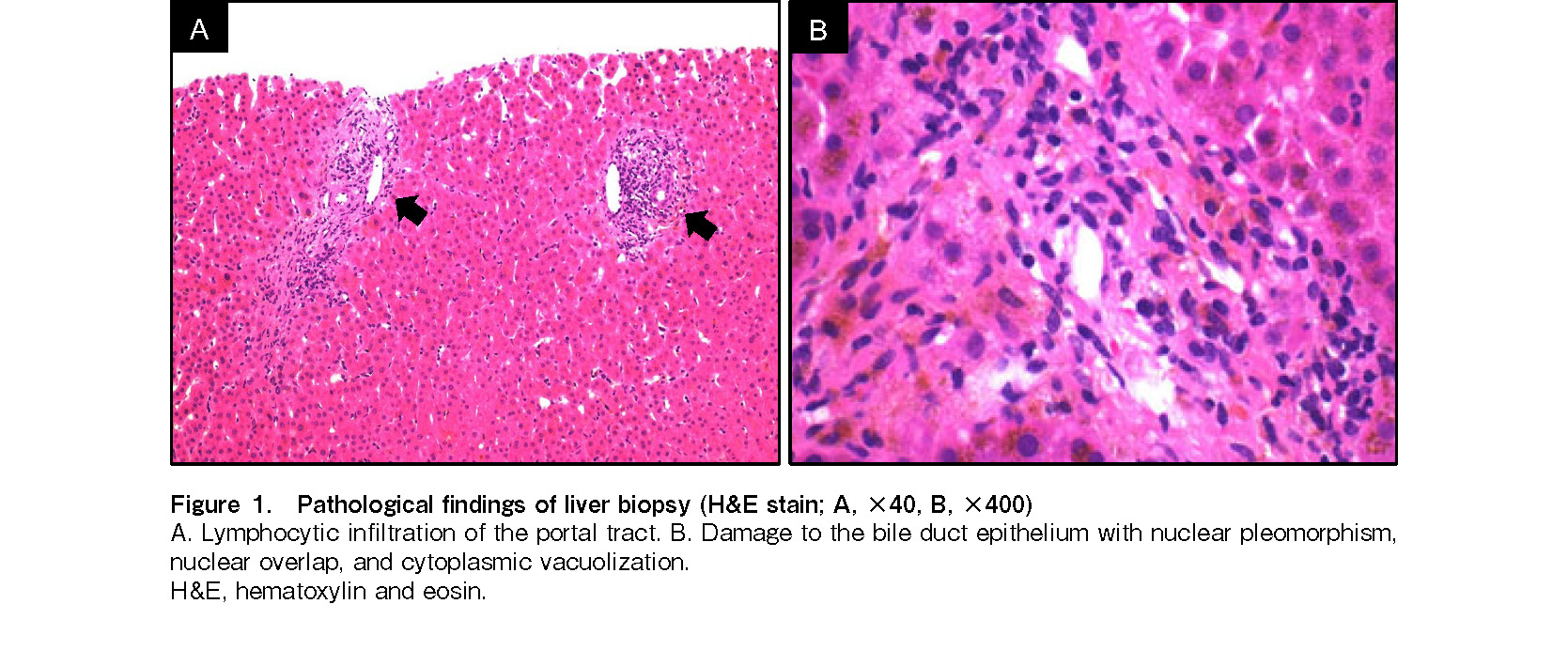
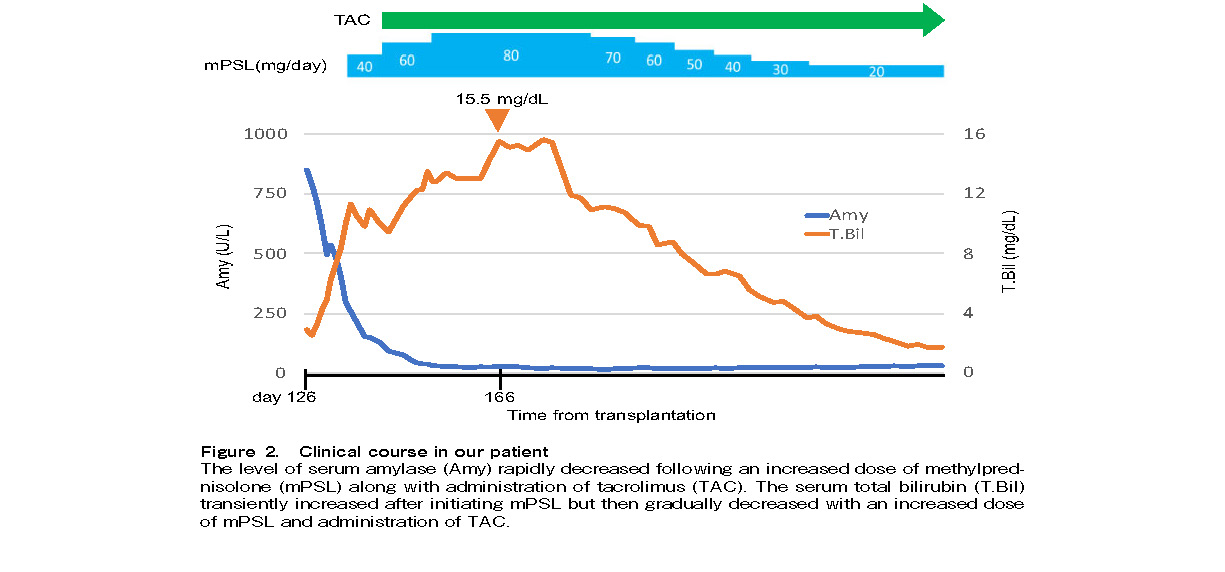
On day 127 (March 2019), the patient complained of upper abdominal pain without any back pain and was admitted to our hospital. Abdominal examination revealed tenderness in the right upper quadrant without rebound. No Murphy's sign or hepatosplenomegaly was detected. Laboratory results showed elevated aspartate aminotransferase (AST, 544 U/L), alanine aminotransferase (ALT, 1,414 U/L), amylase (827 U/L, predominantly pancreatic), and lipase (1,922 U/L) levels. She denied any history of alcohol consumption or drug use. Peripheral blood cytomegalovirus antigenemia test and bacterial cultures were negative. Liver ultrasonography showed only intrahepatic cholestasis, without gallbladder wall thickening or cholangiectasis. Abdominal computed tomography (CT) and magnetic resonance imaging (MRI) scans showed no evidence of acute pancreatitis. A bone marrow biopsy showed 100% donor chimerism and was negative for malignancy, which made it unlikely that her pancreatitis was due to extramedullary ALL relapse. A diagnostic ultrasound-guided liver biopsy was performed, which demonstrated lymphocytic infiltration of the portal tract and bile duct epithelial damage with nuclear overlap and pleomorphism (Figure 1). Therefore, the patient was diagnosed with acute pancreatitis associated with late-onset liver aGVHD. She was treated with mPSL in combination with TAC, maintaining a therapeutic plasma level of 10-15 ng/mL. Her serum amylase level improved rapidly, but the total bilirubin level transiently increased, reaching a maximum of 15.5 mg/dL (maximal aGVHD stage 4, overall grade IV) on day 166. Thereafter, the total bilirubin level gradually decreased, the mPSL dose was slowly tapered, and pancreatitis and liver aGVHD completely resolved (Figure 2).
Discussion
aGVHD is a life-threatening complication of allo-HCT. According to the National Institutes of Health consensus criteria4, aGVHD is subclassified based on the timing of presentation. Classic aGVHD occurs within the first 100 days following allo-HCT, whereas late-onset aGVHD occurs >100 days after allo-HCT4. In our patient, the liver aminotransferase and total bilirubin levels were elevated on day 119 after allo-HCT; therefore, we classified it as late-onset aGVHD.
The total bilirubin level continued to increase until day 166 after allo-HCT, despite treatment with mPSL combined with TAC. This clinical scenario meets some criteria for steroid-refractory (SR) aGVHD, defined as either lack of response to initial treatment, progression during steroid treatment, or prolonged glucocorticoid dependence5. However, liver aGVHD often responds slowly, and further immune suppression often increases the complications. Following treatment with 1-2 mg/kg/day of mPSL combined with TAC, the patient's symptoms improved, her serum amylase level gradually decreased, and she did not develop any signs of synthetic hepatic dysfunction, or symptoms of aGVHD. Therefore, we did not administer any additional therapies for steroid-refractory aGVHD such as rabbit anti-thymocyte globulin, alemtuzumab, or extracorporeal photopheresis.
Liver aGVHD typically presents as elevated levels of AST, ALT, alkaline phosphatase, and total bilirubin. The differential diagnosis of liver disease after allo-HCT includes aGVHD, viral infection, sinusoidal obstructive syndrome, and primary disease infiltration6. Therefore, a liver biopsy is required to achieve a definitive diagnosis. The histopathological findings of liver aGVHD include biliary epithelial damage such as nuclear pleomorphism, loss of nuclear polarity, nuclear overlap, cytoplasmic vacuolization, eosinophilic changes in the cytoplasm, and infrequently, apoptosis. Lymphocytic infiltration of the portal tract may also be observed6. Our patient's diagnosis of late-onset acute liver GVHD was confirmed using an ultrasound-guided biopsy.
It is unlikely that a second allo-HCT or the use of a matched unrelated donor (MUD) PBSC graft increased the risk of aGVHD in our patient. The cumulative incidence of grade II-IV aGVHD after the second allo-HCT is 34%, which is similar to that after the first allo-HCT7. In addition, the incidence of aGVHD in the first 100 days after MUD PBSC transplantation is approximately 50%, which is similar to that following bone marrow-derived grafts8. Based on these studies, we concluded that the second allo-HCT with a PBSC graft did not significantly increase the risk of aGVHD in our patient.
Autopsy-based studies have found a high frequency of asymptomatic pancreatitis following allo-HCT9. In contrast to asymptomatic pancreatitis, symptomatic acute pancreatitis is associated with the use of immunosuppressive drugs1, cytomegalovirus infection10, gallstones, young age, and a history of donor lymphocyte infusion2. Two reports suggest that liver aGVHD may be a major risk factor for acute pancreatitis after allo-HCT9, 10. In addition, Sajiki et al. showed that the development of grade II-IV aGVHD was an independent risk factor for acute pancreatitis following allo-HCT in pediatric patients11. Previous case reports have shown that corticosteroids and calcineurin inhibitors are effective in treating acute pancreatitis, thought to be secondary to aGVHD12,13. Similarly, our patient's acute pancreatitis rapidly responded to mPSL and TAC.
To our knowledge, this is the first successful treatment of acute pancreatitis associated with late-onset liver aGVHD after a second allo-HCT. Prior literature suggests that acute pancreatitis accompanied by liver aGVHD may be overlooked. If practitioners are presented with a similar clinical scenario, we recommend obtaining a diagnostic liver biopsy, unless contraindicated, and prioritizing aGVHD-directed therapy.
Author Contributions
YH and TT diagnosed, treated the patient, and wrote the manuscript. RO provided instructions for the pathological diagnosis. DP and KI provided instructions for writing this article. MH, RS, AY, KA,
Funding Sources
This work was supported by JSPS KAKENHI Grant-in-Aid for Scientific Research (C) Grant Number JP20K08704 (TT), the Japanese Society of Hematology Research Grant (TT), Takeda Science Foundation Research Grant (TT), The Hope from Harper St. Baldrick's Foundation Fellowship (DP), and Hyundai Hope on Wheels Young Investigator Grant (DP).
Ethical Approval and Informed Consent
Written informed consent for publication of the clinical course and any accompanying images was obtained from the patient. This study was approved by the Institutional Review Board (IRB) of Yamagata University Faculty of Medicine and the protocols used in the study were approved by Yamagata University Faculty of Medicine, 2021-S-99.
Conflicts of Interest
The authors declare no conflict of interest. Disclosure forms provided by the authors are available on the website.
References
1.Nieto Y, Russ P, Everson G, Bearman SI, Cagnoni PJ, Jones RB, et al. Acute pancreatitis during immunosuppression with tacrolimus following an allogeneic umbilical cord blood transplantation. Bone Marrow Transplant. 2000; 26: 109-11.
2.Wang XL, Han W, Zhao P, Liu X, Wang JZ, Wang FR, et al. Incidence, Risk Factors, Outcomes, and Risk Score Model of Acute Pancreatitis after Allogeneic Hematopoietic Stem Cell Transplantation. Biol Blood Marrow Transplant. 2020; 26: 1171-8.
3.Akpek G, Valladares JL, Lee L, Margolis J, Vogelsang GB. Pancreatic insufficiency in patients with chronic graft-versus-host disease. Bone Marrow Transplant. 2001; 27: 163-6.
4.Vigorito AC, Campregher PV, Storer BE, Carpenter PA, Moravec CK, Kiem HP, et al. Evaluation of NIH consensus criteria for classification of late acute and chronic GVHD. Blood. 2009; 114: 702-8.
5.Toubai T, Magenau J. Immunopathology and biology-based treatment of steroid-refractory graft-versus-host disease. Blood. 2020; 136: 429-40.
6.Matsukuma KE, Wei D, Sun K, Ramsamooj R, Chen M. Diagnosis and differential diagnosis of hepatic graft versus host disease (GVHD). J Gastrointest Oncol. 2016; 7(Suppl 1): S21-31.
7.Nagler A, Labopin M, Dholaria B, Finke J, Brecht A, Schanz U, et al. Second allogeneic stem cell transplantation in patients with acute lymphoblastic leukaemia: a study on behalf of the Acute Leukaemia Working Party of the European Society for Blood and Marrow Transplantation. Br J Haematol. 2019; 186: 767-76.
8.Anasetti C, Logan BR, Lee SJ, Waller EK, Weisdorf DJ, Wingard JR, et al. Peripheral-blood stem cells versus bone marrow from unrelated donors. N Engl J Med. 2012; 367: 1487-96.
9.Ko CW, Gooley T, Schoch HG, Myerson D, Hackman RC, Shulman HM, et al. Acute pancreatitis in marrow transplant patients: prevalence at autopsy and risk factor analysis. Bone Marrow Transplant. 1997; 20: 1081-6.
10.Salomone T, Tosi P, Raiti C, Stanzani M, Leopardi G, Miglio F, et al. Clinical relevance of acute pancreatitis in allogeneic hemopoietic stem cell (bone marrow or peripheral blood) transplants. Dig Dis Sci. 1999; 44: 1124-7.
11.Sajiki D, Yamashita D, Maemura R, Kitazawa H, Sakaguchi H, Yoshida N, et al. Acute pancreatitis following allogeneic hematopoietic stem cell transplantation in children. Int J Hematol. 2021; 114: 494-501.
12.De Singly B, Simon M, Bennani J, Wittnebel S, Zagadanski AM, Pacault V, et al. [Prolonged acute pancreatitis after bone marrow transplantation]. Gastroenterol Clin Biol. 2008; 32: 413-6.
13.Fernandes SR, Alves AT, Cortes MB, Cortez-Pinto H. Graft-versus-host disease: unexpected presentation with simultaneous hepatitis and pancreatitis. BMJ Case Rep. 2016; 2016: bcr2016215940.
Search
News