Volume 9 (2026) Issue 1 No.4 Pages 21-24
Dear Editor
A study published in the August issue of Blood Cell Therapy has shown significantly higher non-relapse mortality (NRM) after myeloablative (MAC) allogeneic hematopoietic cell transplant (allo-HCT) in South Asians, compared to other Asians and whites older than 45 years1. Allo-HCT is the only curative option for intermediate to adverse-risk and relapsed acute myeloid leukemia (AML). While it is challenging for older individuals to find a human leukocyte antigen (HLA)-matched family donor or an unrelated donor in Indian donor registries, most patients have a haploidentical (haplo) child, making haplo-HCT a time-efficient and economically feasible option in India2. Studies from India on outcomes after haplo-HCT in younger patients have shown higher non-relapse mortality (NRM) rates ranging from 30% to 40%3,4. There is a gap in the outcome data for elderly Indians (≥ 55 years), as most studies have not addressed this issue, and only a limited number of centers offer haplo-HCT to the elderly patient.
After review and approval of the Institutional Ethical Committee (IEC code: 2025-253-IMP-EXP-66) with waiver of informed consent, we retrospectively analyzed the early outcomes following haplo-HCT in elderly patients (chronological age ≥ 55 years) with myeloid malignancies. Patient demographics and clinical parameters were obtained retrospectively from electronic medical records and follow-up notes. All data were collected up to September 30, 2025.
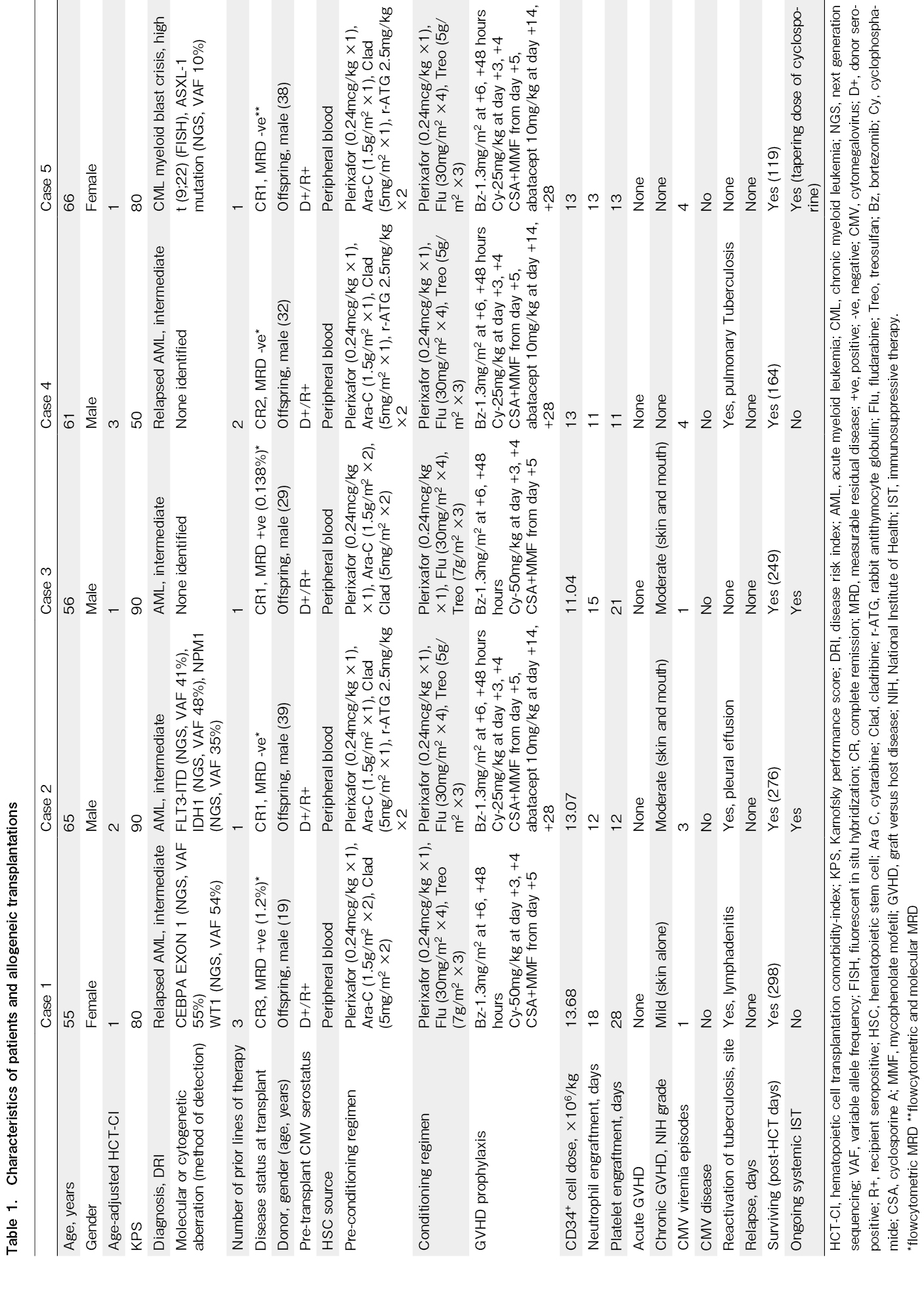
Between December 2024 and May 2025, four patients with AML and one patient with de novo chronic myeloid leukemia (CML) in myeloid blast crisis, aged 55 years or older, underwent haplo-HCT at our institution. The peripheral blood stem cell graft was obtained from an HLA haploidentical son for each patient. The characteristics of the patients, stem cell donors, the allo-HCT procedures, and outcomes are summarized in Table 1.
Patients aged between 55 and 59 years received sequential reduced intensity conditioning (RIC) regimen (preconditioning: plerixafor 0.24 microgram/kg via subcutaneous route at 10 hours before cytarabine on day
Neutrophil and platelet engraftment was achieved in all patients at a median time of 13 days (range, 11-18 days) and 13 days (range, 11-28 days), respectively. None of the patients developed grade 3-4 regimen-related toxicity. The chimerism analysis, performed using the short tandem repeat method on bone marrow aspirates at day +30 and peripheral blood samples at days +60 and +90, revealed 100% donor chimerism in all patients. The bone marrow was in complete morphologic remission, and flow cytometric measurable residual disease was undetectable in all patients at both day +30 and day +90. At a median follow-up of 249 days (range, 119-298 days), none of the patients developed acute GVHD, and two patients developed moderate chronic GVHD (National Institute of Health grading criteria) involving the skin and mouth. Weekly monitoring of cytomegalovirus (CMV) and Epstein-Barr virus (EBV) was performed by quantitative polymerase chain reaction, starting on day +7 and continuing till day +100. Although asymptomatic CMV viremia was detected in all patients, all viremia episodes responded to preemptive therapy with valganciclovir. None of the patients developed EBV reactivation. Three patients developed reactivation of Mycobacterium tuberculosis (M.tuberculosis) at a median time of 90 days (range, 80-140 days) after HCT and are currently receiving antitubercular therapy. Until the last date of data collection, none of the patients experienced disease relapse after HCT, and all are alive.
The leading causes of treatment failure after a melphalan-based RIC-haplo-HCT and PTCy-based GVHD prophylaxis in elderly patients (age ≥ 55 years) with AML are relapse and NRM. The day +100 NRM with this regimen was 21%5. A study by Shima et al. suggested that performance status is a predictor of survival outcome after geriatric allo-HCT (age ≥ 60 years)6. Notably, in our study, three out of five patients had a KPS < 90. A report on haplo-HCT in dyskeratosis congenita has demonstrated the feasibility and safety of a low-dose treosulfan-based RIC regimen in a patient genetically vulnerable to regimen-related toxicity7. Mobilization of leukemic stem cells using plerixafor during MAC for allo-HCT in AML is safe8. All patients in our cohort tolerated the plerixafor-containing low-dose treosulfan-based regimen without grade 3-4 regimen-related toxicity. We incorporated plerixafor to mobilize and sensitize the leukemic stem cells to the conditioning chemotherapy. However, a longer follow-up and a larger sample size are required to assess the efficacy of this strategy in preventing relapse. The absence of acute GVHD and extensive chronic GVHD potentially reflects the efficacy of our GVHD prophylaxis. The safety and effectiveness of short-course bortezomib-based GVHD prophylaxis have been demonstrated in HLA-mismatched unrelated donor RIC transplantation9. The combination of abatacept with PTCy and sirolimus has shown excellent GVHD-free survival after haplo-HCT in non-malignant diseases10. Our abatacept-containing GVHD prophylaxis arm differed in two key aspects: first, it included an intermediate-dose PTCy, and second, the abatacept course was shorter. Notably, three out of five patients developed reactivation of M. tuberculosis. The probable explanations for the higher incidence of tubercular reactivation in this cohort are the high endemicity of tuberculosis in India and impaired cellular immunity after haplo-HCT.
Despite the limitations of retrospective data, a small sample size, and a shorter follow-up period, our study provides real-world data from a low and middle-income country (LMIC) on outcomes during the most vulnerable period after haplo-HCT in the elderly.
In conclusion, our study demonstrates the feasibility and safety of haplo-HCT in elderly patients with myeloid malignancies living in LMICs. Plerixafor, combined with low-dose treosulfan-based conditioning, is well tolerated. The addition of bortezomib and abatacept to the PTCy-based GVHD prophylaxis is feasible and potentially effective.
Acknowledgments
Grammarly software was used for language editing.
Author Contributions
SSR and CP contributed to the concept and design of the study. SSR and BV analyzed the data. SSR, CP, and BV drafted the original manuscript. SSR, CP, BV, MKS, MS, DC, KR, RG, and RK contributed to patient care. All authors critically reviewed and approved the final version of the manuscript.
Conflicts of Interest
The authors declare no conflict of interest. Disclosure forms provided by the authors are available on the website.
References
1.Mohapatra S, Mourad YRA, M Cherniawsky H, Chung SS, Forrest DL, Kaila G, et al. Racial Disparity in Myeloablative Hematopoietic Cell Transplantation Outcomes in Patients with Hematological Malignancies Older Than 45 Years. Blood Cell Ther. 2025; 8: 228-33.
2.Batra A, Perumal Kalaiyarasi J, Kannan K, Mehra N, Ganesan P, Karunakaran P, et al. Haploidentical Hematopoietic Stem Cell Transplantation in Leukemia's: Experience from a Cancer Center in India. Indian J Hematol Blood Transfus. 2021; 37: 463-71.
3.Garg A, Trivedi M, Raj A, Shah K, Patel K, Vachhani A, et al. Haploidentical Stem Cell Transplantation: Half Match but More Hope!―Single Centre Experience from Western India. Indian J Hematol Blood Transfus. 2024; 40: 385-91.
4.Lad DP, Chhabra P, Kasudhan KS, Kaundal S, Chopra M, Singh C, et al. GVHD relapse-free survival after peripheral blood hematopoietic cell transplantation for hematologic malignancies. Blood Cell Ther. 2023; 6: 66-71.
5.Ciurea SO, Shah MV, Saliba RM, Gaballa S, Kongtim P, Rondon G, et al. Haploidentical Transplantation for Older Patients with Acute Myeloid Leukemia and Myelodysplastic Syndrome. Biol Blood Marrow Transplant. 2018; 24: 1232-6.
6.Shima T, Takigawa K, Utsumi S, Yoshino T, Naganuma M, Minami M, et al. Outcomes of allogeneic stem cell transplantation for patients with hematologic diseases ≥60 years old. Blood Cell Ther. 2023; 6: 30-41.
7.Roy SS, Mallik M, Khadwal A, Gogulamudi PKR, Jain A, Prakash G, et al. Haploidentical Hematopoietic Cell Transplantation in Dyskeratosis Congenita with Myelodysplastic Syndrome/Acute Myeloid Leukemia. Blood Cell Ther. 2025; 8: 186-9.
8.Michelis FV, Hedley DW, Malhotra S, Chow S, Loach D, Gupta V, et al. Mobilization of Leukemic Cells Using Plerixafor as Part of a Myeloablative Preparative Regimen for Patients with Acute Myelogenous Leukemia Undergoing Allografting: Assessment of Safety and Tolerability. Biol Blood Marrow Transplant. 2019; 25: 1158-63.
9.Koreth J, Stevenson KE, Kim HT, McDonough SM, Bindra B, Armand P, et al. Bortezomib-based graft-versus-host disease prophylaxis in HLA-mismatched unrelated donor transplantation. J Clin Oncol. 2012; 30: 3202-8.
10.Jaiswal SR, Agarwal M, Bhagawati G, Das BC, Baligar P, Garg M, et al. Long-Term Follow-Up of Abatacept, Post-Transplantation Cyclophosphamide, and Sirolimus-Based Haploidentical Transplantation in Younger Patients with Nonmalignant Diseases. Transplant Cell Ther. 2024; 30: 605.e1-e13.
Search
Actions
News