Volume 9 (2026) Issue 1 No.3 Pages 17-20
Abstract
This report describes a rare pediatric case of seronegative neuromyelitis optica spectrum disorder (NMOSD) following a second haploidentical hematopoietic stem cell transplantation (HSCT) for relapsed acute myeloid leukemia (AML). The neurological syndrome, featuring simultaneous optic neuritis and myelitis, emerged alongside classic chronic graft-versus-host disease (GVHD) manifestations (lichenoid oral changes, arthritis) on day +145 post-transplant. The diagnosis of seronegative NMOSD was established based on clinical and MRI findings, after exclusion of infectious etiologies and absence of aquaporin-4 IgG antibodies in blood and cerebrospinal fluid. Corticosteroids were avoided due to the risk of AML relapse. Instead, plasma exchange, immunotherapy with rituximab and cyclophosphamide, followed by intravenous immunoglobulin maintenance induced rapid neurological improvement and parallel resolution of GVHD symptoms. This case underscores the central nervous system as a target for alloimmunity and presents a successful steroid-sparing treatment strategy for NMOSD manifested after allogenic HSCT.
Introduction
A severe autoimmune inflammatory condition of the central nervous system, Neuromyelitis Optica Spectrum Disorder (NMOSD) is characterized by distinct episodes of optic neuritis, myelitis, and/or brainstem encephalitis. While a majority of cases are seropositive for anti-aquaporin-4 (AQP4-IgG) antibodies, which are detected in both serum and cerebrospinal fluid (CSF), a significant seronegative subset is also recognized. This seronegative status is defined by the absence of not only AQP4-IgG but also antibodies against myelin oligodendrocyte glycoprotein (MOG-IgG). The therapeutic paradigm for NMOSD consists of acute attack management with potent immunosuppression and plasma exchange (PLEX) followed by sustained immunomodulatory therapy for relapse prevention1–3. While high-dose immunosuppression and autologous hematopoietic stem cell transplantation (HSCT) are considered for refractory cases4–6, allogeneic (allo)-HSCT ― though potentially curative for malignant hematological diseases ― itself carries a risk of triggering severe immune-mediated complications, including de novo NMOSD7–12. The rare occurrence of NMOSD after allo-HSCT for unrelated indications suggests a shared alloimmune pathway with GVHD, prompting questions about serological characteristics, timing of onset, presentation, and therapeutic response. We describe a pediatric case of seronegative NMOSD following haploidentical HSCT and its association with chronic GVHD.
Clinical Case
A 14-year-old female patient was managed at the State Healthcare Institution
Due to the high risk of relapse and the lack of time to identify a fully HLA-matched unrelated donor, the decision was made to proceed with an HSCT using T-cell replete peripheral stem cells from a haploidentical, ABO-mismatched donor. The conditioning regimen consisted of treosulfan (42 g/m2), fludarabine (150 mg/m2), and etoposide (60 mg/kg). The graft composition was as follows: CD34+ cells 5.7×106/kg, CD3+ cells 290×106/kg. GVHD prophylaxis was performed with post-transplantation cyclophosphamide (Pt-Cy) (50 mg/kg/day on days +3 and +4), followed by tacrolimus (until day+100) and mycophenolate mofetil (MMF) (until day+30).
The early post-transplant course was notable for the absence of significant infectious or immunological complications. Neutrophil engraftment was achieved on day +18 and platelet engraftment on day+25. Clinical and hematological MRD-negative remission with 100% donor chimerism was confirmed on days+30, +60, +90, and +180 post-transplant.
The patient was subsequently followed on an outpatient basis with standard supportive care. Upon tacrolimus taper, she developed mild acute skin GVHD (grade 1), which was successfully managed with topical therapy. On day+258, pancytopenia developed, and subsequent investigations confirmed a hematological relapse of AML. A fludarabine, cytarabine, idarubicin (FLAI) re-induction course failed to achieve remission, with bone marrow blasts persisting at 56%, expressing BCL2 and CD38.
As salvage therapy, a second T-cell replete HSCT from an alternative ABO-mismatched haploidentical donor was performed using a conditioning regimen, which was consisted of a FLA course (fludarabine, cytarabine) augmented with venetoclax (up to 400 mg/day) and daratumumab (16 mg/kg), followed by treosulfan (42 g/m2) and etoposide (60 mg/kg). The unmanipulated graft contained CD34+ cells 13.9×106/kg and CD3+ cells 610×106/kg. GVHD prophylaxis was identical to the first transplant (Pt-Cy, tacrolimus, MMF). Neutrophil engraftment occurred on day+17, with full hematological recovery, MRD-negative status, and 100% donor chimerism. Two consolidation cycles of 5-azacytidine (35 mg/m2 for 5 days) were administered for remission maintenance. The early post-transplant period was again uncomplicated; MMF was discontinued on day+30 and tacrolimus on day+100.
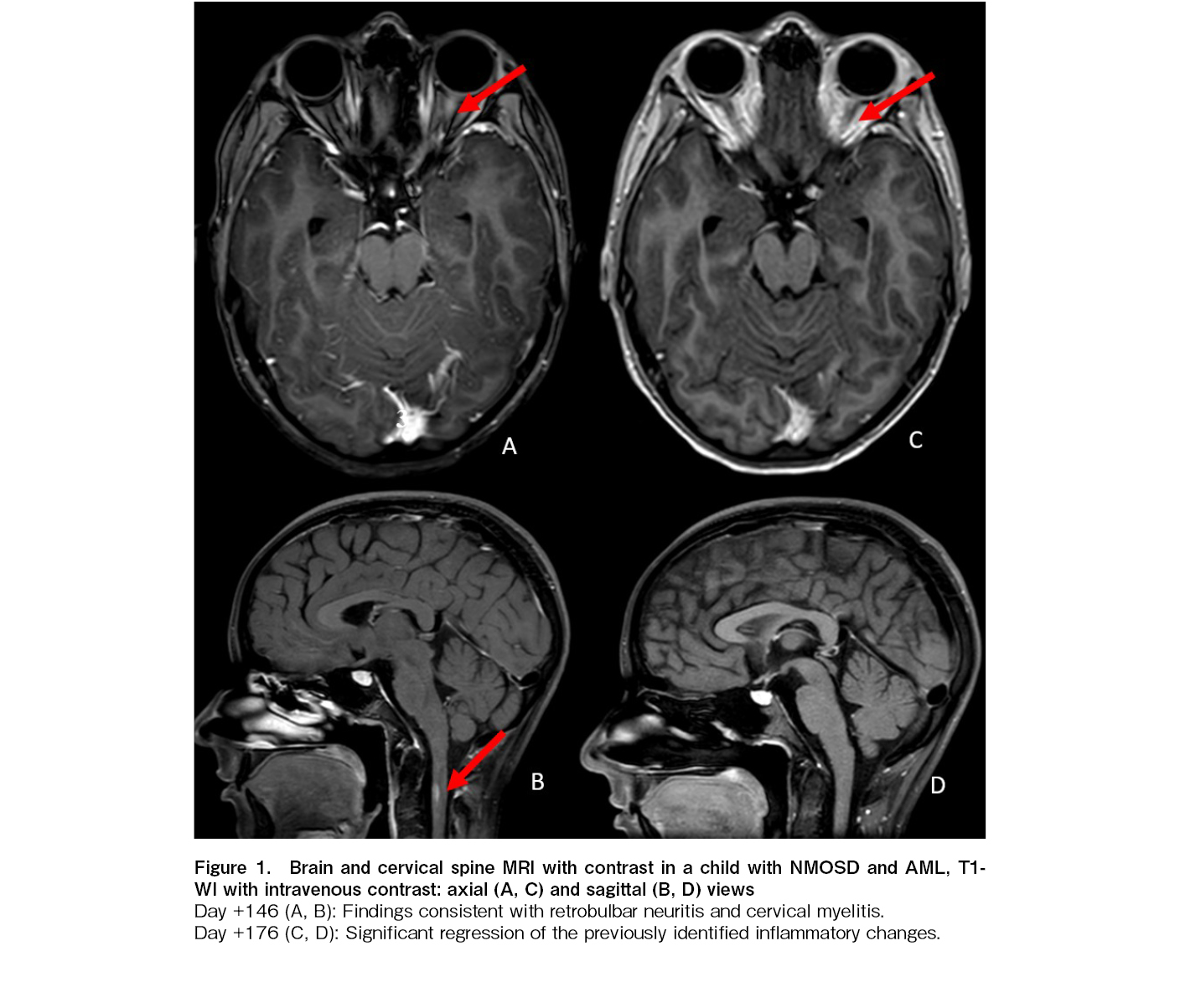
On day+145 after the second HSCT, the patient reported pain in the left orbit upon eye movement. Concurrently, she developed arthralgia and exhibited lichenoid changes of the oral mucosa, findings highly characteristic of chronic GVHD. Ophthalmological examination revealed unilateral papilledema. A contrast-enhanced MRI of the brain showed findings consistent with isolated retrobulbar neuritis of the left optic nerve and a focal myelitic lesion at the C2 level. No other demyelinating lesions were detected on spinal cord MRI. Cerebrospinal fluid (CSF) analysis was negative for viral pathogens [cytomegalovirus (CMV), epstein-barr virus (EBV), human herpesvirus 6 (HHV6), varicella-zoster virus (VZV), herpes simplex virus (HSV1/2), BK polyomavirus (BK), JC polyomavirus (JCV), Parvovirus B19] and blast cells; a mild elevation in CSF protein was noted. AQP4-IgG antibodies and MOG-IgG were not detected in the serum or CSF. A diagnosis of seronegative NMOSD, likely of alloimmune origin, was established.
The patient received sequential therapy with four sessions of PLEX, two doses of rituximab (375 mg/m2, administered two weeks apart), and one pulse of cyclophosphamide (750 mg/m2). Treatment response was evaluated after this induction phase, which led to a significant improvement and complete resolution of the orbital pain. Maintenance therapy consisted of six monthly courses of intravenous immunoglobulin (IVIG) at 2 g/kg per course. A follow-up MRI on day+176 demonstrated a partial reduction of inflammatory changes in the optic nerve and complete resolution of the spinal cord lesion (Figure 1). The finding paralleled by the complete resolution of arthralgia and significant improvement in oral lichenoid manifestations.
A third relapse of AML was diagnosed on day+468 after the second HSCT. Notably, the patient exhibited no further signs or symptoms of neuromyelitis optica reactivation thereafter.
Discussion
The management of NMOSD is predicated on a dual strategy: acute attack therapy with high-dose corticosteroids, often supported by therapeutic PLEX or IVIG in refractory cases, and long-term remission maintenance using immunosuppressants or targeted agents1–3,14. The development of seronegative NMOSD post-HSCT, presenting with simultaneous optic neuritis and myelitis, represents a severe and rare neurological complication. In this case, its concurrence with classic chronic GVHD features underscores a probable alloimmune origin, positioning it as a severe neurological form of GVHD. This presentation aligns with previously reported, though heterogeneous, cases of post-transplant central nervous system autoimmunity, such as isolated optic neuritis (Ooi et al.7), steroid-refractory seronegative neuromyelitis optica requiring plasmapheresis (Baumer et al.9), and devastating AQP4-IgG-positive NMOSD (Diamanti et al.10). A pivotal finding was the parallel evolution of NMOSD with systemic chronic GVHD (oral lichenoid changes and arthritis), a clinical synergy that strongly supports the concept of the central nervous system as a definitive locus for alloimmune aggression, akin to the
In the present case, corticosteroid therapy was deliberately avoided due to concerns regarding potential provocation of an AML relapse. The syndrome demonstrated striking sensitivity to aggressive therapy, including PLEX, rituximab, and cyclophosphamide, which resulted in significant clinical improvement and provides a successful management blueprint for such severe post-HSCT cases. This rapid and positive neurological response not only confirms the immune-mediated pathogenesis but also highlights the therapeutic efficacy of B-cell depletion and immunomodulation. The sustained neurological remission despite subsequent AML relapse further underscores the specificity of this immunosuppressive approach. Crucially, the simultaneous resolution of both NMOSD and systemic GVHD manifestations under therapy demonstrates a clear clinical association and suggests a common pathogenic link between these conditions.
Author Contributions
GZ wrote the manuscript and treated the patient. NV performed neurological management, monitoring, and contributed to the patient's therapy. GO served as the head of the department.
Informed Consent
An informed consent was obtained from the patient's parents for publication of this case report.
Conflicts of Interest
The authors declare no conflict of interest. Disclosure forms provided by the authors are available on the website.
References
1.Jarius S, Aktas O, Ayzenberg I, Bellmann-Strobl J, Berthele A, Giglhuber K, et al. Update on the diagnosis and treatment of neuromyelitis optica spectrum disorders (NMOSD) – revised recommendations of the Neuromyelitis Optica Study Group (NEMOS). Part I: Diagnosis and differential diagnosis. J Neurol. 2023; 270: 3341-68.
2.Palace J, Leite MI, Jacob A. A practical guide to the treatment of neuromyelitis optica. Pract Neurol. 2012; 12: 209-14.
3.Abboud H, Petrak A, Mealy M, Sasidharan S, Siddique L, Levy M. Treatment of acute relapses in neuromyelitis optica: Steroids alone versus steroids plus plasma exchange. Mult Scler. 2016; 22: 185-92.
4.Greco R, Bondanza A, Vago L, Moiola L, Rossi P, Furlan R, et al. Allogeneic hematopoietic stem cell transplantation for neuromyelitis optica. Ann Neurol. 2014; 75: 447-53.
5.Skorobogatova EV, Kirgizov KI, Volkova EY, Piliya SV, Blagonravova OL, Pristanskova EA, et al. Efficacy of high-dosage therapy for autoimmune diseases of the CNS in children: the experience of a multidisciplinary group on the study of efficacy and late effects. Pediatriya – Zhurnal im G.N. Speranskogo. 2019; 98: 214-20. (in Russian)
6.Sharrack B, Saccardi R, Alexander T, Badoglio M, Burman J, Farge D, et al. Autologous haematopoietic stem cell transplantation and other cellular therapy in multiple sclerosis and immune-mediated neurological diseases: updated guidelines and recommendations from the EBMT Autoimmune Diseases Working Party (ADWP) and the Joint Accreditation Committee of EBMT and ISCT (JACIE). Bone Marrow Transplant. 2020; 55: 283-306.
7.Ooi J, Takahashi S, Tajika K, Tajo A, Tani K, Asano S. Immune-mediated optic neuritis after unrelated allogeneic bone marrow transplantation. Blood. 1998; 91: 2619.
8.Stefanou MI, Bischof F. Central and peripheral nervous system immune-mediated demyelinating disease after allogeneic hematopoietic stem cell transplantation. J Neuroimmunol. 2017; 307: 74-81.
9.Baumer FM, Kamihara J, Gorman MP. Neuromyelitis optica in an adolescent after bone marrow transplantation. Pediatr Neurol. 2015; 52: 119-24.
10.Diamanti L, Franciotta D, Berzero G, Bini P, Farina LM, Colombo AA, et al. Late post-transplant anti-aquaporin-4 Ab-positive optic neuritis in a patient with AML. Bone Marrow Transplant. 2015; 50: 1125-6.
11.Hümmert MW, Stadler M, Hambach L, Gingele S, Bredt M, Wattjes MP, et al. Severe allo-immune antibody-associated peripheral and central nervous system diseases after allogeneic hematopoietic stem cell transplantation. Sci Rep. 2021; 11: 8527.
12.Deng RX, Wu YJ, Xu LP, Liu KY, Huang XJ, Zhang XH. Clinical risk factors and prognostic model for idiopathic inflammatory demyelinating diseases after haploidentical hematopoietic stem cell transplantation in patients with hematological malignancies. Am J Hematol. 2021; 96: 1407-19.
13.Kumar P, Chaudhary B, Devi S, Dubey G, Chatterjee A, Sharma D, et al. Therapeutic applications and strategies of neurobiomarkers in neurological disorders: A narrative review. NeuroMarkers. 2025; 2: 100113.
14.Pittock SJ, Barnett M, Bennett JL, Berthele A, de Sèze J, Levy M, et al. Ravulizumab in Aquaporin-4-Positive Neuromyelitis Optica Spectrum Disorder. Ann Neurol. 2023; 93: 1053-68.
Search
Actions
News