Volume 9 (2026) Issue 1 No.2 Pages 9-16
Abstract
Antiemetic therapy is an essential component of supportive care following hematopoietic stem cell transplantation (HSCT). Chemotherapy and irradiation used in conditioning regimens frequently induce severe nausea and vomiting, which can significantly impair patients' quality of life. Although recent guidelines recommend a triple combination of a 5-hydroxytryptamine-3 (5-HT3) receptor antagonist, dexamethasone, and an neurokinin 1 (NK1) receptor antagonist, antiemetic practices vary widely across countries and regions. This study aimed to investigate current antiemetic policies among the Asia-Pacific Blood and Marrow Transplantation (APBMT) centers.
A web-based questionnaire survey using SurveyMonkey was distributed via email from the APBMT office between December 7, 2021, and January 21, 2022. The survey addressed antiemetic strategies used in HSCT conditioning regimens. Responses were received from 28 centers across 14 countries.
Among the participating centers, 93% reported that physicians were primarily responsible for antiemetic decision-making, with limited involvement from pharmacists or multidisciplinary teams. The most commonly used conditioning regimens for allogeneic HSCT were busulfan and cyclophosphamide (Bu-CY) (72%) and fludarabine and busulfan (Flu-Bu) (62%), whereas high-dose melphalan (83%) and carmustine (BCNU), etoposide, cytarabine arabinoside, and melphalan (BEAM) (69%) were predominant for autologous HSCT. Despite guidelines recommending olanzapine as an additional antiemetic in highly emetogenic chemotherapy, its routine implementation remains limited, even in high-risk settings. Notably, dexamethasone is frequently avoided in allogeneic HSCT, likely due to concerns about its immunosuppressive effects. The incidence of vomiting varied, with 36% of centers reporting rates of 10% or higher, even among those with institutional antiemetic policies.
In conclusion, this survey highlighted substantial variation in antiemetic strategies across the APBMT centers. The limited use of olanzapine reflects ongoing concerns regarding its side effects, while the frequent avoidance of dexamethasone in allogeneic HSCT represents a deviation from current guideline recommendations. Given the complexity of HSCT and the varying side effect profiles of antiemetic agents, a multidisciplinary approach to treatment planning, including that of pharmacists and dietitians, could optimize supportive care. Future prospective studies are warranted to evaluate the safety, efficacy, and feasibility of olanzapine- and steroid-sparing antiemetic strategies to improve patient outcomes.
Introduction
From a nutritional management perspective, the appropriate use of antiemetic agents is essential to ensure sufficient oral intake and maintain the nutritional status of patients during treatment1. In addition, adequate antiemetic control plays a key role in preserving enteral feeding tubes throughout the transplant period. Without sufficient control of nausea and vomiting, episodes of emesis may result in the dislodgement of nasogastric feeding tubes. In the context of allogeneic hematopoietic stem cell transplantation (HSCT), structured nutritional support has been recognized as a key factor in improving clinical outcomes2. This is particularly important in HSCT, where intensive chemotherapy and irradiation are used in conditioning regimens, which frequently lead to severe nausea and vomiting. These symptoms not only reduce patients' quality of life but also impair oral nutritional intake. Therefore, effective management of chemotherapy-induced nausea and vomiting (CINV) is crucial for supporting both nutritional status and overall treatment outcomes.
Recent guidelines have emphasized the importance of combination antiemetic therapy for the management of CINV. A triple regimen consisting of a 5-hydroxytryptamine-3 (5-HT3) receptor antagonist, dexamethasone, and an neurokinin 1 (NK1) receptor antagonist is currently recommended for patients undergoing HSCT3–5. Olanzapine, an antipsychotic with potent antiemetic properties, is considered an optional, but beneficial addition, particularly for managing both acute and delayed phases of CINV4. Despite these recommendations, antiemetic practices vary widely across the globe, highlighting the limited guidance available for individualized prophylaxis in HSCT settings6. A recent comprehensive literature review by Jordan et al. found that NK1 receptor antagonists were underutilized in many studies, despite their demonstrated benefit in improving antiemetic efficacy in the HSCT7. Contributing factors to these variations include differences in local medical practices, drug availability, economic constraints, and varying interpretations of existing guidelines. Understanding such variability is critical for identifying gaps in current practice and guiding improvements in care. The Asia-Pacific Blood and Marrow Transplantation Group (APBMT) encompasses a diverse range of countries and regions, each with distinct healthcare systems and resources, where the number of HSCT has been steadily rising annually8. To clarify the current policy of antiemetic use in the participating countries and regions of the APBMT, we conducted an international questionnaire survey focused on antiemetic use in HSCT.
Materials and Methods
A web-based questionnaire survey was conducted using SurveyMonkey and distributed via email sent from the APBMT office between December 7, 2021, and January 21, 2022. The draft questionnaire was developed by the Nutrition Working Group of the APBMT and underwent multiple rounds of discussion before being finalized at the APBMT annual congress held in Thailand in 2021. The complete survey questionnaire is provided in the
Results
Responses were collected from 28 centers across 14 countries. Demographic details of the respondents included representatives from Japan (n=7), China (n=3), Australia, India, Iran, Pakistan, Singapore, and Thailand (n=2 each). Single responses were obtained from Indonesia, Korea, Malaysia, Mongolia, Myanmar, and Nepal (n=1 each). Because some centers did not provide complete answers to all survey items due to differences in available medications and the use or omission of TBI, the total number of responses for certain questions did not necessarily add up to 28 centers. The reported annual numbers of allogeneic HSCT in a year were
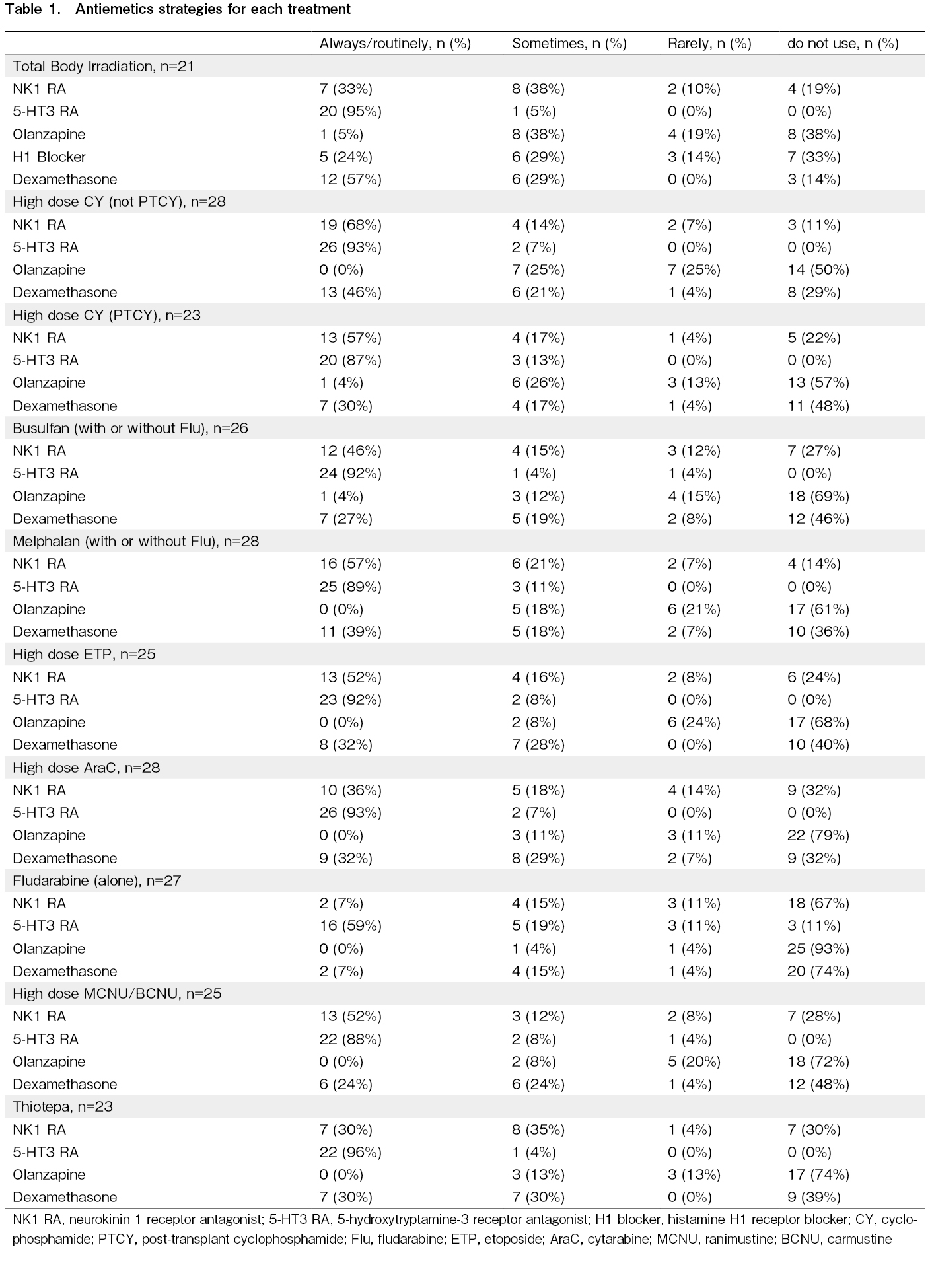
The main results of antiemetics for each treatment are shown in Table 1. While 5-HT3 receptor antagonists are often used as antiemetics, even at minimal to low emetic risk, olanzapine is not routinely used in many hospitals, even at high emetic risk. In addition to differences in the use of olanzapine, several centers reported routinely using alternative agents instead of dexamethasone when administering conditioning regimens. Specifically, one center used diphenhydramine when cyclophosphamide or busulfan was administered. When melphalan or thiotepa was used, one center used diphenhydramine and another used metoclopramide. One center used metoclopramide when etoposide was given, and another used hydrocortisone when cytarabine was administered as part of routine practice, considering its anti-allergic and antiemetic effects.
In seven hospitals, TBI was not used as part of the conditioning regimen. Although TBI is classified as high emetic risk in radiation therapy, and the combination of a 5-HT3 receptor antagonist with dexamethasone is recommended, three respondents reported not using dexamethasone.
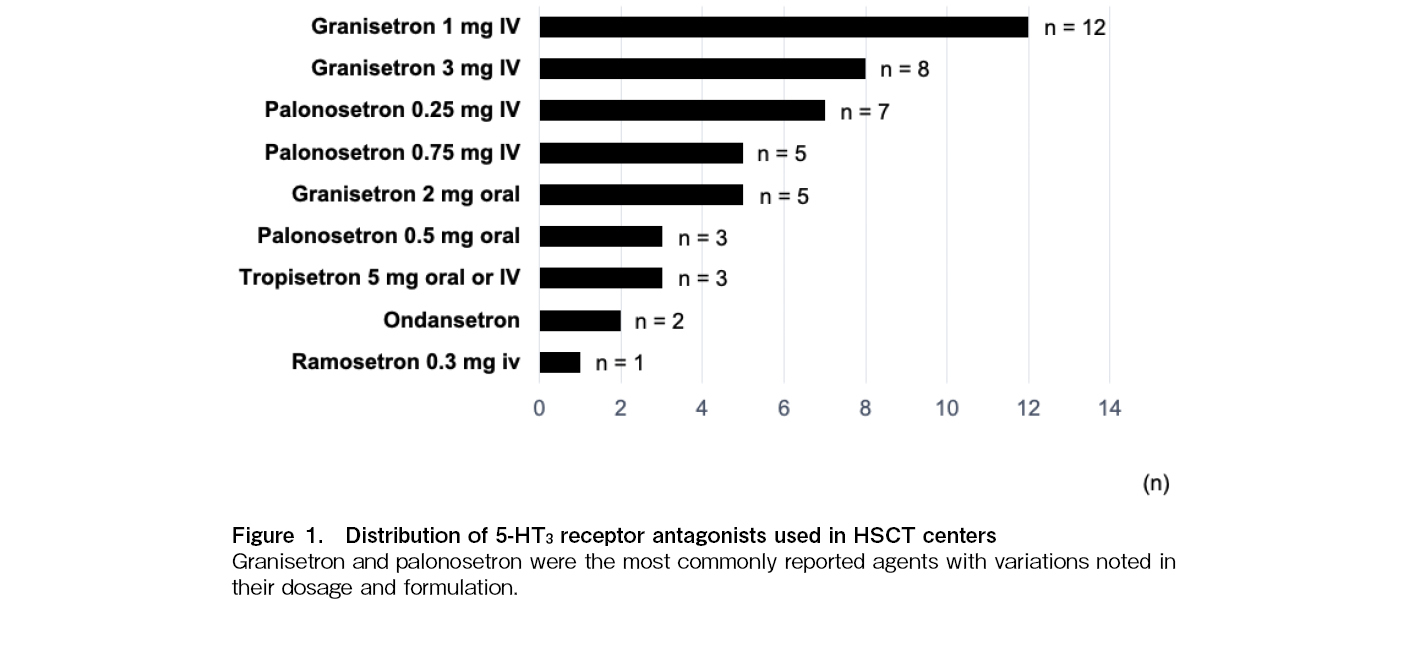
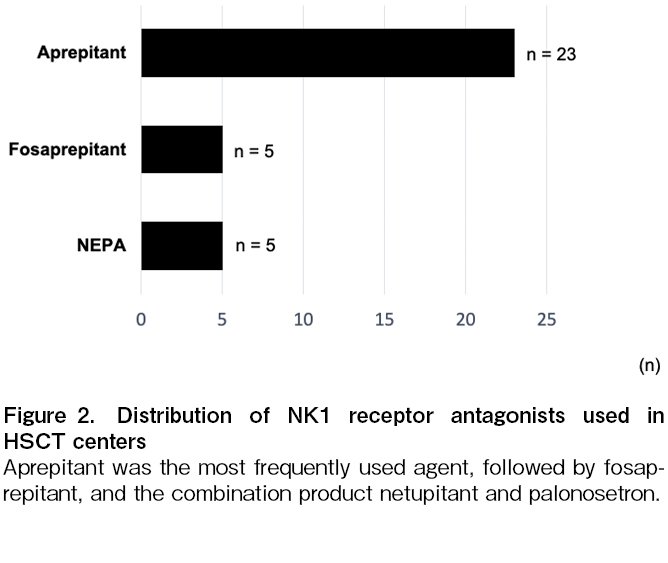
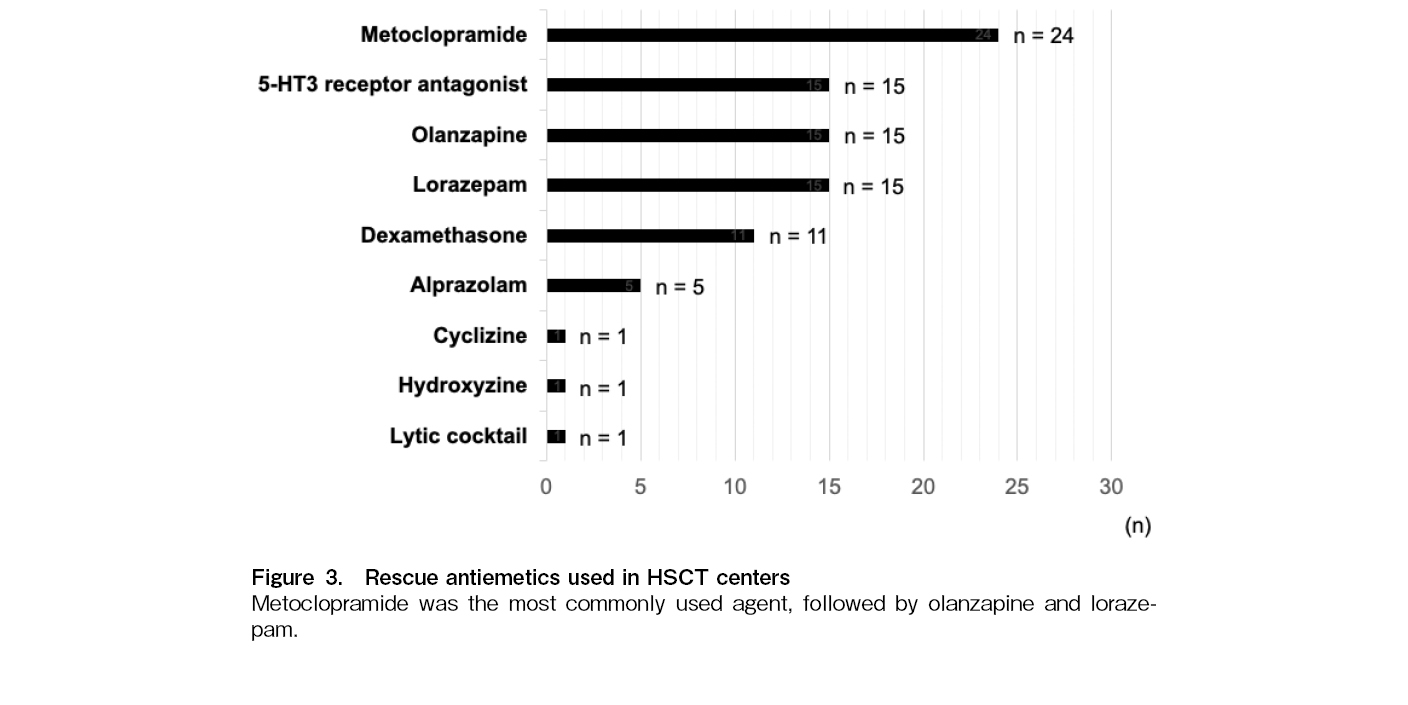
Figure 1 illustrates the distribution of 5-HT3 receptor antagonists used across participating centers, with granisetron and palonosetron being the most commonly reported antagonists. Figure 2 presents the utilization of NK1 receptor antagonists, indicating a strong preference for aprepitant over fosaprepitant and combination of netupitant and palonosetron (NEPA). The survey also assessed the use of rescue antiemetics in HSCT centers (Figure 3). Metoclopramide was the most frequently used agent, followed by 5-HT3 receptor antagonists, olanzapine and lorazepam. Dexamethasone was also reported despite concerns about its immunosuppressive effects in allogeneic HSCT. Less commonly used agents included alprazolam, cyclizine, hydroxyzine, and lytic cocktails containing chlorpromazine, pethidine and promethazine.
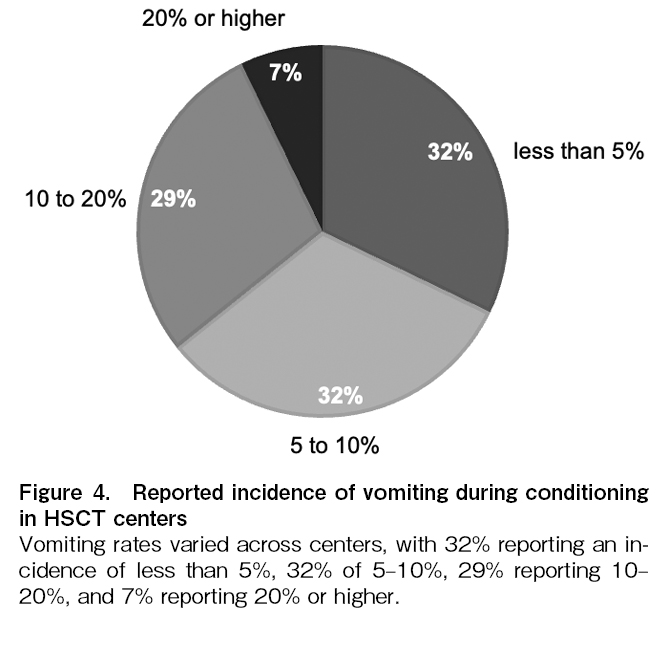
The incidence of vomiting during conditioning regimen administration varied among centers, regardless of the antiemetic strategies employed. Specifically, 32% of centers reported rates below 5%, 32% reported 5-10%, 29% reported 10-20%, and 7% reported rates of 20% or higher (Figure 4).
Discussion
The survey results revealed substantial variability in antiemetic practices across countries and regions within the APBMT. These discrepancies may arise from differences in local clinical practices, medication availability, economic conditions, and interpretations of international guidelines. Similar regional disparities were noted in a previous APBMT international survey on nutritional support following HSCT, which reported considerable variation in caloric targets, initiation criteria for parenteral nutrition, and the use of immunonutrition across centers in the Asia-Pacific region9. These findings reflect broader challenges in standardizing supportive care and highlight the importance of developing region-specific strategies for both antiemetic and nutritional management. In particular, the observed geographical variability likely reflects disparities in healthcare infrastructure and access to essential medications, which may limit the ability of some regions to fully implement guideline-recommended therapies10. Accordingly, there is a clear need for tailored context-specific guidelines that account for local healthcare systems, resource availability, and economic constraints to support equitable and effective patient care.
One of the key findings of this survey is the widespread use of 5-HT3 receptor antagonists, which is consistent with current guidelines recommending these agents as a core component of antiemetic therapy, owing to their established efficacy3–5. In contrast, olanzapine, despite its demonstrated effectiveness, particularly in managing delayed nausea and vomiting in autologous HSCT patients11―was not widely used. Although it is recommended as a fourth agent to augment the standard triplet regimen, concerns regarding sedation, metabolic side effects, and potential psychiatric risks may limit its use, particularly in immunocompromised patients. Nevertheless, these adverse effects are generally mild and not considered major clinical concerns in most cases11,12. However, clinical evidence regarding the safety and efficacy of olanzapine in HSCT remains limited, being confined to only a few prospective studies11,13 and several retrospective single-center reports14–16, and further research is needed to establish its optimal role.
The avoidance of dexamethasone in certain regimens, especially allogeneic HSCT, is another notable finding. Although dexamethasone is effective for managing CINV, its well-documented immunosuppressive properties raise concerns about an increased risk of infections and other steroid-related complications in this vulnerable patient population17. This cautious approach highlights the delicate balance clinicians must maintain between achieving adequate CINV control and minimizing adverse effects, particularly in immunocompromised individuals undergoing HSCT.
The survey also revealed discrepancies in antiemetic practices between autologous and allogeneic HSCT. Patients receiving allogeneic HSCT are often prescribed more potent antiemetic regimens, likely because of the higher intensity of conditioning regimens and the elevated risk of CINV. However, this practice is not applied consistently across centers, indicating a need for more standardized protocols. Antiemetic decision-making is primarily physician-driven, with limited involvement from pharmacists or multidisciplinary teams. Given the complexity of HSCT and the diverse side effect profiles of antiemetic agents, fostering a more collaborative approach to treatment planning may enhance patient outcomes. Involving pharmacists, dietitians, and palliative care specialists can provide valuable expertise in optimizing supportive care, managing adverse effects, and supporting adequate nutritional intake throughout the treatment course.
The variation in the incidence of vomiting among centers suggests that the current prophylactic strategies may not be uniformly effective in all settings. Multiple factors could contribute to these differences, including differences in conditioning regimen intensity, patient characteristics, and institutional antiemetic policies that extend beyond guideline recommendations. The relatively high proportion of centers reporting vomiting rates of 10% or higher (36%) indicates a potential need for more intensive prophylactic approaches in certain cases. Particularly, the limited use of olanzapine, despite its proven efficacy in managing both acute and delayed CINV, may be a contributing factor. Further investigations are warranted to evaluate whether incorporating olanzapine into standard antiemetic regimens can help reduce vomiting rates, especially in centers with higher reported incidence. Additionally, patient-specific factors, such as individual emetogenic risk, prior chemotherapy exposure, and genetic susceptibility to nausea and vomiting, should be explored to personalize antiemetic strategies and improve treatment outcomes.
This study has some limitations. First, as a questionnaire-based survey, it is subject to selection bias, potentially limiting the generalizability of the findings. Second, since the survey responses were provided by individual representatives from each center, they may not fully capture institutional consensus or reflect actual clinical practice. In addition, the incidence of vomiting was not analyzed according to the type or intensity of conditioning regimens, which may have contributed to the variability in reported rates among centers. This lack of stratified analysis represents another methodological limitation of the present study. Third, the cross-sectional nature of the survey precluded evaluation of longitudinal trends in antiemetic practices. Future studies should incorporate a prospective, multi-institutional design to monitor evolving patterns in antiemetic management and assess their impact on clinical outcomes.
In summary, although the survey demonstrated general adherence to the guidelines for the use of 5-HT3 receptor antagonists, it also revealed notable gaps in the utilization of olanzapine and dexamethasone. Addressing these gaps requires further research to validate the safety and efficacy of these agents in HSCT settings, particularly olanzapine. Additionally, integrating patient-reported outcomes, such as satisfaction, quality of life, and treatment preferences, into clinical evaluations of antiemetic practices would be valuable. Consideration of patient perspectives could lead to improved adherence to antiemetic regimens and ultimately enhance overall patient outcomes across diverse healthcare settings.
Despite its proven efficacy, olanzapine is not widely used in HSCT settings. Similarly, while dexamethasone is an effective antiemetic agent, its use is often deliberately avoided in allogeneic HSCT due to concerns about steroid-associated complications in immunocompromised patients. These deviations from guideline-recommended antiemetic regimens highlight the need for individualized approaches in clinical practice. Future prospective studies are warranted to evaluate the safety, efficacy, and feasibility of olanzapine and steroid-sparing antiemetic strategies, with the goal of optimizing symptom control and improving outcomes across diverse healthcare settings.
Author Contributions
KY and SF designed the study, developed the questionnaire and analyzed the results. SF also supervised the overall conduct of the study. JP, AH, TF, HI, JWL, YI, BG, DW, RS, MB, HH, TA, PR, BSP, DS, AAG, OA, CW, GGG, SIy, SIq, MR, AK, JK, JS, JC, DM, and CYX contributed data to the study. SO and MI reviewed and discussed the study design. All authors reviewed and approved the final manuscript.
Conflicts of Interest
MI is affiliated with the Department of Promotion for Blood and Marrow Transplantation at the Aichi Medical University School of Medicine, and the department is endowed by AIR WATER INC.. Other authors declare no conflicts of interest. MI, SO, and MB are one of the editors of Blood Cell Therapy. They were not involved in the editorial evaluation or decision to accept this article for publication.
References
1.Fuji S, Cheng J, Yakushijin K, Wanitpongpun C. Nutritional support in allogeneic hematopoietic stem cell transplantation Asian perspective. Blood Cell Ther. 2022; 5: 54-60.
2.Fuji S, Einsele H, Savani BN, Kapp M. Systematic Nutritional Support in Allogeneic Hematopoietic Stem Cell Transplant Recipients. Biol Blood Marrow Transplant. 2015; 21: 1707-13.
3.Hesketh PJ, Kris MG, Basch E, Bohlke K, Barbour SY, Clark-Snow RA, et al. Antiemetics: American Society of Clinical Oncology Clinical Practice Guideline Update. J Clin Oncol. 2017; 35: 3240-61.
4.Hesketh PJ, Kris MG, Basch E, Bohlke K, Barbour SY, Clark-Snow RA, et al. Antiemetics: ASCO Guideline Update. J Clin Oncol. 2020; 38: 2782-97.
5.Einhorn LH, Rapoport B, Navari RM, Herrstedt J, Brames MJ. 2016 updated MASCC/ESMO consensus recommendations: prevention of nausea and vomiting following multiple-day chemotherapy, high-dose chemotherapy, and breakthrough nausea and vomiting. Support Care Cancer. 2017; 25: 303-8.
6.Báez-Gutiérrez N, Suárez-Casillas P, Pérez-Moreno MA, Blázquez-Goñi C, Abdelkader-Martín L. Antiemetic prophylaxis regimens in haematologic malignancies patients undergoing a hematopoietic stem cell transplantation. Which is the best standard of care? A systematic review. Eur J Haematol. 2024; 113: 564-75.
7.Jordan K, Jordan B, Burgt JV, Jahn F. Exploring antiemetic strategies in hematologic malignancies: a comprehensive literature review and evaluation of antiemetic efficacy in patients receiving high-dose chemotherapy prior to hematopoietic stem cell transplantation. Support Care Cancer. 2025; 33: 622.
8.Iida M, Liu K, Huang XJ, Huang H, Kuwatsuka Y, Moon JH, et al. Report on hematopoietic cell transplantations performed in 2018/2019 focusing on the trends of selection of stem cell sources in the Asia-Pacific region: APBMT Activity Survey. Blood Cell Ther. 2023; 6: 114-23.
9.Fuji S, Mori T, Lee V, Cheng J, Linton N, Lie A, et al. A Multi-Center International Survey Related to the Nutritional Support after Hematopoietic Stem Cell Transplantation Endorsed by the ASIA Pacific Blood and Marrow Transplantation (APBMT). Food and Nutrition Sciences. 2012; 3: 417-21.
10.Okamoto S, Iida M, Hamad N, Duarte FB, Sureda A, Srivastava A, et al. American Society of Transplantation and Cellular Therapy International Affair Committee: Report of the Third Workshop on Global Perspective to Access to Transplantation at the 2022 Tandem Meeting. Transplant Cell Ther. 2023; 29: 410-7.
11.Clemmons AB, Orr J, Andrick B, Gandhi A, Sportes C, DeRemer D. Randomized, Placebo-Controlled, Phase III Trial of Fosaprepitant, Ondansetron, Dexamethasone (FOND) Versus FOND Plus Olanzapine (FOND-O) for the Prevention of Chemotherapy-Induced Nausea and Vomiting in Patients with Hematologic Malignancies Receiving Highly Emetogenic Chemotherapy and Hematopoietic Cell Transplantation Regimens: The FOND-O Trial. Biol Blood Marrow Transplant. 2018; 24: 2065-71.
12.Zhang XL, Ying JE. Olanzapine for the Prevention and Treatment of Chemotherapy-Induced Nausea and Vomiting: A Review to Identify the Best Way to Administer the Drug. Curr Oncol. 2022; 29: 8235-43.
13.Nakagaki M, Barras M, Curley C, Butler JP, Kennedy GA. A randomized trial of olanzapine versus palonosetron versus infused ondansetron for the treatment of breakthrough chemotherapy-induced nausea and vomiting in patients undergoing hematopoietic stem cell transplantation. Support Care Cancer. 2017; 25: 607-13.
14.Trifilio S, Welles C, Seeger K, Mehta S, Fishman M, McGowan K, et al. Olanzapine Reduces Chemotherapy-induced Nausea and Vomiting Compared With Aprepitant in Myeloma Patients Receiving High-dose Melphalan Before Stem Cell Transplantation: A Retrospective Study. Clin Lymphoma Myeloma Leuk. 2017; 17: 584-9.
15.Kato J, Uchida M, Ishikawa M, Tatsuta S, Yoshimi T, Ishida K, et al. Efficacy of intensive antiemetic therapy including olanzapine in multiple myeloma patients treated with high-dose melphalan with autologous stem cell transplantation. Support Care Cancer. 2025; 33: 777.
16.Karpen R, Sen J, Wall S, Musson S, Tossey J. Evaluation of palonosetron, fosaprepitant, and olanzapine as antiemetic prophylaxis for fludarabine and melphalan-based conditioning regimens prior to allogeneic hematopoietic stem cell transplants. Leuk Res. 2024; 136: 107431.
17.Swartz SL, Dluhy RG. Corticosteroids: clinical pharmacology and therapeutic use. Drugs. 1978; 16: 238-55.
Search
Actions
News