Volume 9 (2026) Issue 1 No.5 Pages 25-30
Abstract
Background: Relapsed Hodgkin lymphoma (HL) after autologous stem cell transplantation (auto-HSCT) remains a major therapeutic challenge with few curative options. Allogeneic hematopoietic stem cell transplantation (allo-HSCT) may provide durable disease control, yet outcome data from tertiary-care centres in India are limited.
Objective: To evaluate real-world outcomes of allo-HSCT in patients with chemotherapy-sensitive relapsed HL at a tertiary care centre in North India.
Methods: This retrospective study included patients with chemotherapy-sensitive relapsed HL who underwent allo-HSCT at the Postgraduate Institute of Medical Education and Research (PGIMER), Chandigarh, between January 2017 and December 2023. Clinical data including baseline characteristics, transplant parameters, complications, and survival outcomes were reviewed. Descriptive statistics were used, and overall survival (OS) was estimated using the Kaplan-Meier method.
Results: Five patients (median age 18 years; range 14-30) with chemotherapy-sensitive relapse following auto-HSCT underwent allogeneic transplantation in complete remission (CR) after salvage therapy. Donors included haploidentical (n=3), matched unrelated (n=1), and matched sibling (n=1). Four patients received reduced-intensity conditioning; one received myeloablative conditioning. Graft-versus-host disease (GVHD) prophylaxis included post-transplant cyclophosphamide (PT-Cy) in four patients. Neutrophil and platelet engraftment occurred at median 15 and 16 days, respectively. All patients developed bacterial infections; one succumbed to Gram-negative sepsis. Acute grade III GVHD occurred in one patient, and two developed limited chronic GVHD. At a median follow-up of 44 months, four patients remained in continuous remission, with a 3-year OS of 80%.
Conclusion: In Indian patients with chemotherapy-sensitive relapsed HL, allo-HSCT using reduced-intensity and reduced toxicity conditioning regimens and post-transplant cyclophosphamide-based regimens has proven feasible and effective, yielding encouraging survival outcomes within routine clinical practice.
Introduction
Hodgkin lymphoma (HL) is a highly curable malignancy, with frontline chemotherapy and radiotherapy achieving durable remission in most patients. However, around 10-30% may develop relapsed or refractory disease, often requiring high-dose chemotherapy followed by autologous hematopoietic stem cell transplantation (auto-HSCT) for salvage1. In cases where patients relapse after auto-HSCT, allogeneic hematopoietic stem cell transplantation (allo-HSCT) offers a potentially curative option, leveraging the graft-versus-lymphoma effect2,3. Nevertheless, allo-HSCT carries significant risks, including graft-versus-host disease (GVHD), infections, and transplant-related mortality4,5.
Although global data on allo-HSCT outcomes in HL are available, real-world evidence from India remains limited. Factors such as demographic variability, donor availability, infectious disease burden, and disparities in access to comprehensive supportive care may distinctly impact transplant outcomes in the Indian context. In this retrospective study, we analyse the clinical characteristics, transplant-related outcomes, and survival of patients with HL who underwent allo-HSCT at a tertiary care center in India. Our goal is to provide real-world insights into the efficacy and safety of allo-HSCT in this population.
Methods
A retrospective analysis of institutional practice data was undertaken at the Postgraduate Institute of Medical Education and Research (PGIMER), Chandigarh, India, encompassing medical records of patients with chemotherapy-sensitive relapse following autologous HSCT who underwent allogeneic HSCT during January 2017-December 2023. Eligible patients included those who had previously received frontline chemotherapy with or without radiotherapy, followed by autologous HSCT, and subsequently experienced disease relapse or refractory status, necessitating allo-HSCT with curative intent. Based on donor type, comorbidity profile, and pre-HSCT performance status, patients received either reduced-intensity conditioning (fludarabine (Flu)-cyclophosphamide (Cy)-total body irradiation (TBI): Flu 30 mg/m2 day(d) −6 to −2, Cy 14.5 mg/kg d −6, −5, TBI d −1 (2 Gy ×2), reduced-toxicity myeloablative conditioning (Flu-treosulfan (Treo): Flu 30 mg/m2 d −6 to −2, Treo 12 g/m2 d −6 to −4) or myeloablative conditioning (Flu- busulfan (Bu): Flu 30 mg/m2 d −5 to −2, Bu 3.2 mg/kg d −5 to −2), aiming to optimize the balance between toxicity and disease control. The study aimed to evaluate clinical characteristics, transplant-related variables, and outcomes in this high-risk population.
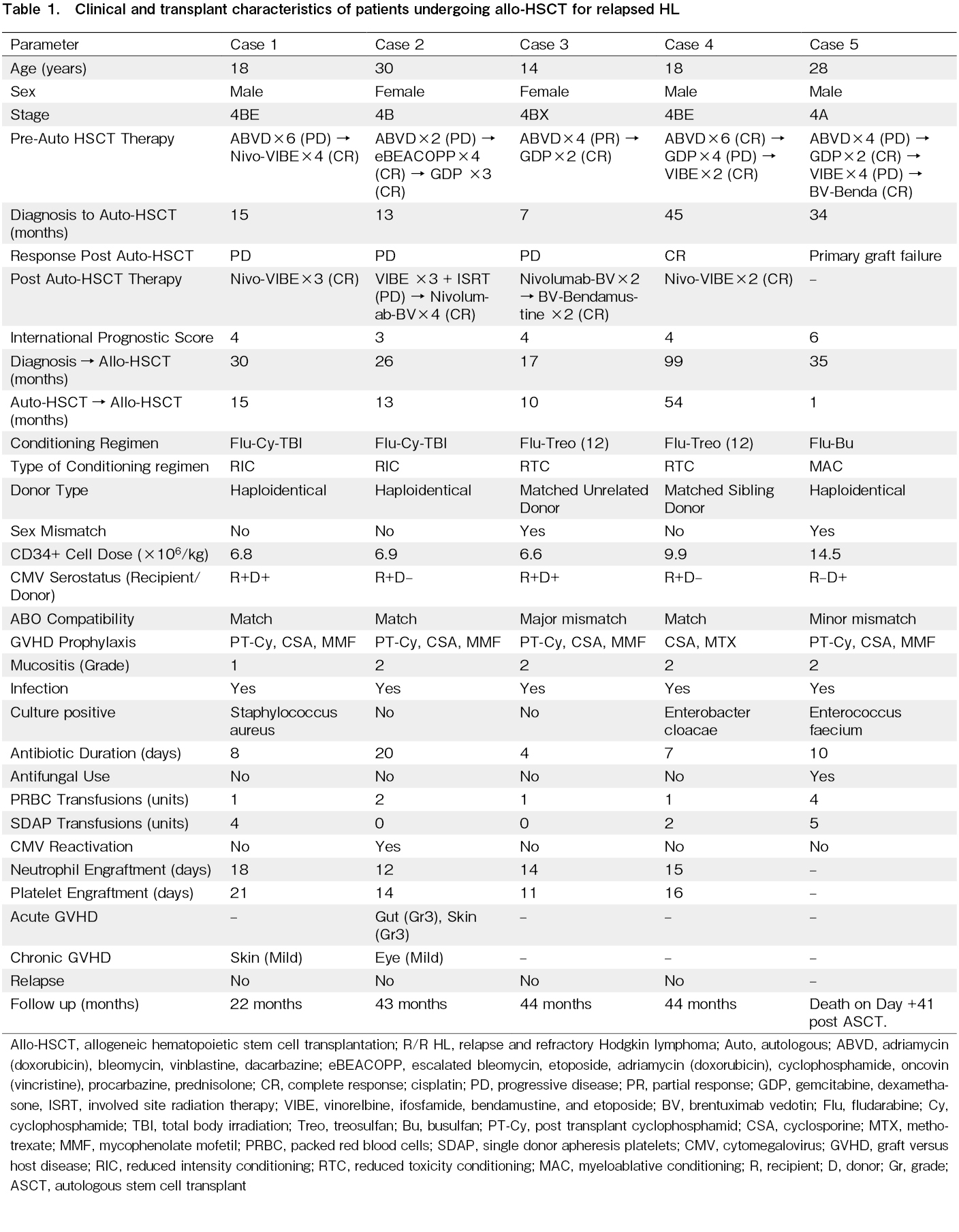
Data collected included baseline patient demographics, disease-related features such as disease stage at diagnosis, International Prognostic Score (IPS), and prior lines of therapy. Transplant-related details comprised donor type (matched sibling, unrelated, or haploidentical), conditioning regimen intensity, and GVHD prophylaxis used. Post-transplant outcomes assessed included neutrophil and platelet engraftment times, incidence and severity of acute and chronic GVHD (graded per NIH criteria), cytomegalovirus (CMV) reactivation, transfusion requirements, infection episodes, disease relapse, and overall survival (OS) (Table 1). Neutrophil engraftment was defined as an absolute neutrophil count (ANC) ≥ 0.5 × 109/L sustained for three consecutive days, while platelet engraftment was defined as ≥ 20 × 109/L without transfusions for at least seven days. A written informed consent was obtained from all the patients in accordance with Declaration of Helsinki. This study was approved by intramural ethics committee (INT-CHMO-774-2025).
Statistical analysis
Descriptive statistics were used to summarize patient characteristics, transplant-related variables, and post-transplant outcomes. Categorical variables were expressed as frequencies and percentages, while continuous variables were presented as mean ± standard deviation (SD) or median with interquartile range (IQR), depending on the data distribution. OS was estimated using the Kaplan-Meier method. Median time to neutrophil and platelet engraftment was calculated. Transplant-related complications, including infections, GVHD, and CMV reactivation, were recorded and analysed descriptively. Due to the limited sample size (n=5), no formal statistical tests or multivariate analyses were performed. Statistical analysis was performed using the Statistical Package for the Social Sciences (SPSSⓇ), version 20.0 (SPSS Inc., IBM Corp).
Results
Five patients with chemotherapy-sensitive relapse following ASCT underwent allo-HSCT between 2017-2023. The median age at allo-HSCT was 18 years (range 14-30), with a male-to-female ratio of 3:2. All patients had advanced-stage disease (Stage IV), and most had high-risk disease based on the International Prognostic Score, ranging from 3 to 6. All patients had previously undergone auto-HSCT, with allo-HSCT performed upon relapse or graft failure. At the time of allo-HSCT, all patients were in complete remission (CR) after salvage therapy given after relapse post auto-HSCT. The median interval between the two transplants was 13 months (range: 1-54 months).
The donor types included haploidentical donors (n=3), a matched sibling donor (MSD; n=1), and a matched unrelated donor (MUD; n=1). Among patients who had received PD-1 inhibitors before allo-HSCT, the median washout interval between last dose and conditioning was 53 days. Case 1 had Nivo-VIBE (nivolumab 40mg d 1, vinorelbine 25 mg/m2 d 1, ifosfamide 5 g/m2 d 1, 2, Bendamustine 90 mg/m2 d 1, 2, and etoposide 100 mg/m2 d 1 to 3) with a washout of 60 days, Case 2 received nivolumab 40mg d 1 and brentuximab vedotin (BV) 1.8 mg/kg d 1 with 45 days, Case 3 received nivolumab-BV with 35 days, and Case 4 received Nivo-VIBE with 90 days. Conditioning regimens administered were Flu-Cy-TBI; n=2, Flu-Treo; n=2, and Flu-Bu; n=1. GVHD prophylaxis included post-transplant cyclophosphamide (PT-Cy), cyclosporine (CSA), and mycophenolate mofetil (MMF) in four patients, while one patient with an MSD received CSA and methotrexate. Neutrophil engraftment occurred at a median of 15 days (range: 12-18), and platelet engraftment at a median of 16 days (range: 11-21). Transfusion requirements varied across patients, with packed red blood cells transfusions ranging from 1 to 4 units, and platelet support from 0 to 5 single donor apheresis units.
All five patients developed bacterial infections during the peri-engraftment phase, with culture positivity in three (Staphylococcus aureus, Enterobacter cloacae, and Enterococcus faecium). One patient (Case 5) experienced primary graft failure following auto-HSCT despite an adequate infused CD34+ cell dose (8.2 × 106/kg), > 90% cell viability, and no cryopreservation issues, with heavy pre-treatment considered the likely cause. He subsequently underwent allo-HSCT on d 31 post-auto-HSCT but succumbed to multidrug-resistant Gram-negative sepsis on d 10 post-allo-HSCT (d 41 post-auto-HSCT). The indication for allo-HSCT in this case was graft failure, leading to early transplant-related mortality. CMV reactivation occurred in one haploidentical HSCT recipient and was managed successfully with intravenous ganciclovir. No other viral or fungal reactivations were documented. One patient developed grade 3 acute GVHD involving the gut and skin, which resolved with systemic corticosteroids. Two patients experienced mild chronic GVHD (skin and ocular involvement), both of which responded after use of topical steroids and calcineurin inhibitors. At a median follow-up of 44 months (range 22-44), four patients remained in long-term remission, yielding a three-year OS of 80%, with all survivors relapse-free. Vaccination has been completed in all patients except one, in whom live vaccines are still pending.
Discussion
Allo-HSCT continues to be a potentially curative treatment for eligible patients with chemotherapy-sensitive relapsed HL following failure of auto-HSCT. Our small real-world series demonstrates encouraging outcomes using reduced intensity and reduced toxicity conditioning regimens in an low-and middle-income countries (LMIC) setting, with a 3-year OS of 80%, aligning with results from larger international cohorts despite the challenges inherent to a heavily pretreated population2,6. These findings highlight the feasibility and effectiveness of allo-HSCT in selected patients with advanced HL, particularly when supported by optimized modern supportive care and individualized transplant strategies.
Our transplantation approach incorporated matched related, matched unrelated, and haploidentical donors. Three of the five patients underwent haploidentical transplantation, reflecting the growing reliance on haploidentical donors in the Indian context, where matched sibling or unrelated donors are often unavailable, and highlighting the increasing accessibility and effectiveness of haploidentical HSCT with post-transplant PT-Cy4. The conditioning regimens included two reduced-intensity (Flu-Cy-TBI) and two reduced-toxicity myeloablative (Flu-Treo) fludarabine-based protocols, while one patient received a myeloablative Flu-Bu4 regimen. Reduced intensity conditioning (RIC) and reduced toxicity conditioning (RTC) regimens have become increasingly favored in the management of chemotherapy-sensitive relapsed HL, as they offer reduced non-relapse mortality while achieving sustained disease control in a subset of patients3,5. The observed graft-versus-lymphoma effect appears to play a crucial role in this context, especially when chemotherapy resistance limits further cytotoxic therapy options7. GVHD remains a central concern in allogeneic transplantation, given its impact on morbidity and long-term quality of life. In our series, the incidence of clinically significant GVHD was relatively low: one patient developed grade III acute GVHD, and two had limited chronic GVHD. This likely reflects the GVHD-sparing effect of PT-Cy in haploidentical transplants, which is now widely recognized for its dual benefit of GVHD prevention and retention of graft-versus-leukemia activity8. Engraftment kinetics (median neutrophil recovery: 15 days; platelet recovery: 16 days) were comparable to published data, reinforcing the feasibility of these protocols in our setting9.
A significant challenge in our cohort was the high incidence of early infectious complications. All five patients experienced bacterial infections during the peri-engraftment period requiring intravenous antibiotics, with three cases confirmed by positive cultures. This high incidence reflects the profound immunosuppression post-allo-HSCT, compounded by India's high burden of infections10. One patient succumbed to Gram-negative sepsis before achieving neutrophil recovery. Infection-related mortality continues to be a significant barrier in transplant centers across LMICs, where environmental exposure to resistant pathogens, limited access to intensive supportive care, and constrained infrastructure compound the risks11. CMV reactivation occurred in one patient, a common complication in allograft recipients12. Pre-emptive monitoring and antiviral therapy remain essential in preventing its clinical impact.
Our single-centre series demonstrating a 3-year OS of 80% after allo-HSCT for chemotherapy-sensitive relapsed HL is consistent with outcomes from larger multicentre and registry studies, despite the challenges of an LMIC setting. In the prospective study by Das-Gupta et al., transplant-naïve high-risk patients achieved a similar 3-year OS of 80.7%, underscoring the benefit of allo-HSCT in carefully selected cohorts1. Ahmed et al. highlighted the impact of RIC and RTC regimen choice on outcomes, a finding mirrored in our experience, where the use of Flu-Cy-TBI and Flu-Treo regimens supported reliable engraftment and durable remissions2. The EBMT registry analysis by Martínez et al. confirmed haploidentical HSCT with PT-Cy as a viable alternative to MSD or MUD, with lower chronic GVHD rates, mirroring our favourable experience with three haploidentical transplants3. Spanish protocols likewise established the feasibility of RIC allo-HSCT in relapsed HL4. Collectively, these studies contextualize our findings, demonstrating that with judicious patient selection, RIC or RTC conditioning, and PT-Cy prophylaxis, Indian outcomes can parallel those in high-income countries, even amid higher infection burdens.
While our findings are encouraging, the study has several limitations. The small sample size and retrospective nature limit the generalizability of the results and preclude statistical comparisons or identification of prognostic factors. Furthermore, the lack of a comparator group, such as patients treated with chemo-immunotherapy alone restricts our ability to evaluate the relative benefit of allo-HSCT in this setting. Despite these limitations, our findings provide valuable real-world insights into the feasibility of RIC and RTC regimens for allo-HSCT in Indian patients with chemotherapy-sensitive relapsed HL.
Conclusion
Allo-HSCT offers durable remission in chemotherapy-sensitive relapsed HL post auto-HSCT; however, outcomes in chemo-refractory disease may differ and require further study. While challenges such as infections and GVHD persist, careful patient selection, haploidentical donor use, and PT-Cy-based prophylaxis offer a feasible pathway to success. These real-world insights contribute to the growing evidence supporting allo-HSCT in HL and underscore the need for tailored approaches in diverse healthcare environments.
Conflicts of Interest
The authors declare no conflict of interest. Disclosure forms provided by the authors are available on the website. DL is one of the editors of Blood Cell Therapy. He was not involved in the editorial evaluation or decision to accept this article for publication.
Acknowledgments
We are grateful to our patients for their participation in the study.
Author Contributions
SW, RNS, AS, CS, and AJ wrote the manuscript. AJ, GP, PM, and AK contributed to the study design. MUS and RH performed laboratory investigations. PM and AK reviewed the manuscript and gave final approval.
References
1.Connors JM, Cozen W, Steidl C, Carbone A, Hoppe RT, Flechtner HH, et al. Hodgkin lymphoma. Nat Rev Dis Primers. 2020; 6: 61.
2.Das-Gupta E, Thomson KJ, Bloor AJC, Clark AD, Mackinnon S, Kayani I, et al. Allo-HSCT in transplant-naïve patients with Hodgkin lymphoma: a single-arm, multicenter study. Blood Adv. 2019; 3: 4264-70.
3.Ahmed S, Ghosh N, Ahn KW, Khanal M, Litovich C, Mussetti A, et al. Impact of type of reduced-intensity conditioning regimen on the outcomes of allogeneic haematopoietic cell transplantation in classical Hodgkin lymphoma. Br J Haematol. 2020; 190: 573-82.
4.Martínez C, Gayoso J, Canals C, Finel H, Peggs K, Dominietto A, et al. Post-Transplantation Cyclophosphamide-Based Haploidentical Transplantation as Alternative to Matched Sibling or Unrelated Donor Transplantation for Hodgkin Lymphoma: A Registry Study of the Lymphoma Working Party of the European Society for Blood and Marrow Transplantation. J Clin Oncol. 2017; 35: 3425-32.
5.Sureda A, Canals C, Arranz R, Caballero D, Ribera JM, Brune M, et al. Allogeneic stem cell transplantation after reduced intensity conditioning in patients with relapsed or refractory Hodgkin's lymphoma. Results of the HDR-ALLO study – a prospective clinical trial by the Grupo Español de Linfomas/Trasplante de Médula Osea (GEL/TAMO) and the Lymphoma Working Party of the European Group for Blood and Marrow Transplantation. Haematologica. 2012; 97: 310-7.
6.Ge S, Lepic K, Bhindi R, Berg T, Khalaf D, Leber B, et al. Outcomes of Allogeneic Stem Cell Transplant in Patients with Relapsed/Refractory Hodgkin Lymphoma. Curr Oncol. 2025; 32: 118.
7.Robinson SP, Sureda A, Canals C, Russell N, Caballero D, Bacigalupo A, et al. Reduced intensity conditioning allogeneic stem cell transplantation for Hodgkin's lymphoma: identification of prognostic factors predicting outcome. Haematologica. 2009; 94: 230-8.
8.Lofthouse M. GVT effect of reduced-intensity allogeneic transplantation for Hodgkin's lymphoma. Nat Rev Clin Oncol. 2005; 2: 432.
9.Robinson SP, Sureda A, Canals C, Russell N, Caballero D, Bacigalupo A, et al. Reduced intensity conditioning allogeneic stem cell transplantation for Hodgkin's lymphoma: identification of prognostic factors predicting outcome. Haematologica. 2009; 94: 230-8.
10.Lad DP, Malhotra P, Khadwal A, Prakash G, Ray P, Mishra B, et al. Infections Are a Major Cause of Prolongation of Hospital Stay in Hematopoietic Stem Cell Transplants in Tropical, Developing Countries: Profile of a Transplant Center in North India. Blood. 2014; 124: 5853.
11.Saharman YR, Karuniawati A, Severin JA, Verbrugh HA. Infections and antimicrobial resistance in intensive care units in lower-middle income countries: a scoping review. Antimicrob Resist Infect Control. 2021; 10: 22.
12.Hakki M, Riddell SR, Storek J, Carter RA, Stevens-Ayers T, Sudour P, et al. Immune reconstitution to cytomegalovirus after allogeneic hematopoietic stem cell transplantation: impact of host factors, drug therapy, and subclinical reactivation. Blood. 2003; 102: 3060-7.
Search
Actions
News