Volume 9 (2026) Issue 1 No.6 Pages 31-40
Abstract
Background: Acute gastrointestinal graft-versus-host disease (GI-GVHD) is a complication of allogeneic hematopoietic stem cell transplantation (HSCT) that requires early diagnosis. Fecal calprotectin (FC) is a biomarker of intestinal inflammation. We studied the role of FC in differentiating GI-GVHD from other diarrhea.
Methods: We prospectively studied allogeneic HSCT recipients between 2017 and 2020. FC levels were measured pre-transplant, on post-transplant day 14, on day 1 of diarrhea, and post-steroid treatment (day 3-7 of steroid treatment).
Results: We studied 116 patients with median age 14.5 (5-29) years; 84 were male (72.4%). Fifty-seven patients (49.1%) developed diarrhea post-HSCT, 34 (59.7%) patients had GI-GVHD, and 23 patients (40.3%) developed other diarrheal illnesses. FC level on day1 of diarrhea among GI-GVHD patients (n=33) was higher (63 μg/g [Q1-Q3:25.6-358.5]) compared to other diarrhea (27.5 μg/g [21.1-60.4], p=0.045). FC cut-off > 53.7 μg/g had sensitivity (78.6%) and specificity (57.9%) with area under the receiver operating characteristic curve of 0.67 to predict GI-GVHD on day 1 of diarrhea. FC levels in steroid non-responders (n=7) were higher (311.5 μg/g [40.5-1,291.5]) than responders (n=11) (31.2 μg/g [20.8-137.2] μg/g); p=0.03. Haplomatch, severe GI-GVHD, and coexisting skin GVHD were significant predictors of poor treatment response.
Conclusion: A higher FC value on day 1 of diarrhea aids in the diagnosis of acute GI-GVHD and predicts poor response to treatment.
Introduction
Acute graft versus host disease (GVHD) affects 40%-60% of allogeneic hematopoietic stem cell transplantation (HSCT) recipients, usually affecting the skin, gastrointestinal (GI) tract, and liver1. The GI tract is the most commonly affected organ by GVHD2. Gastrointestinal GVHD (GI-GVHD) manifests with diarrhea; however, other symptoms of nausea, vomiting, abdominal pain, and bleeding per rectum have also been described. In post-transplant patients presenting with diarrhea, distinguishing gastrointestinal graft-versus-host disease (GI-GVHD) from infection or mucositis is challenging. GVHD mortality depends on the response to initial steroid therapy; mortality ranges from 49% (steroid-resistant) to 27% (steroid-responsive)3. Early diagnosis of GI-GVHD and prediction of steroid response will help in risk stratification, upgrade to second-line therapy, and reduce GI-GVHD-related mortality4.
Various blood and stool biomarkers have been studied to assist in the diagnosis of diarrheal illnesses. Fecal calprotectin (FC) is a neutrophil breakdown protein, which has been used clinically to detect gut inflammation and differentiate inflammatory bowel disease (IBD) from irritable bowel syndrome (IBS) (elevated in IBD). FC performed better than the other biomarkers in differentiating IBD from IBS5. FC has also been used to follow up the disease status of patients with IBD following initiation of therapy. There are studies that assessed the use of FC in allogeneic HSCT settings in relation to GI-GVHD and found that the FC level was higher among patients with GI-GVHD, and higher levels of FC correlated with poor steroid response6. However, these findings need to be validated in Asian countries. It also remains to be studied whether pre-transplant or pre-symptomatic (post-transplant) FC can be used to predict future GI-GVHD.
Previous studies on this subject were conducted in Europe, and the largest cohort included 95 patients6–8. To address these issues, our study analyzed a larger cohort of patients from Asia, first to validate the role of FC in the diagnosis of GI-GVHD in an Indian context (where FC levels are known to be higher even in healthy individuals9) and also to investigate new objectives such as predicting the onset of GI-GVHD using FC (pre-transplant and during asymptomatic post-transplant period).
Materials and Methods
We prospectively studied patients who underwent allogeneic HSCT at a tertiary care center in South India between 2017 and 2020. This study was approved by the Institutional Review Board and Ethics Committee (IRB min. No. 10394 dated 3rd Nov 2016). All patients included in the study provided informed written consent for publication of study data.
This study aimed to evaluate the utility of FC levels in aiding the diagnosis of acute GI-GVHD in patients with diarrhea in a post-HSCT setting. The primary objective of this study was to determine whether day 1 diarrhea FC levels can differentiate between GI-GVHD and other causes of diarrhea. The secondary objectives included: to measure the difference in FC levels between mild (grade 1 and grade 2) and severe (grade 3 and grade 4) clinical GI-GVHD grades, to measure the difference between day 1 of diarrhea FC levels between steroid responders and steroid non-responders, and whether FC levels before diarrhea onset can predict forthcoming GI-GVHD. These objectives should enhance the early diagnosis, severity assessment, and treatment planning for GI-GVHD using FC.
Study participants
Patients who underwent allogeneic HSCT for various indications and consented to participate in the study with informed consent and assent (for children) were included.
Study procedures
Stool samples for FC were collected prior to conditioning chemotherapy in all included patients. Subsequently, stool samples were collected for FC analysis on post-transplant day 14. Patients who developed diarrhea after transplantation were evaluated for GI-GVHD and other causes of diarrhea. Stool cultures for bacteria, stool samples for parasitic ova/cysts, cytomegalovirus (CMV) titers in blood, and CMV PCR from rectal tissue were sent for evaluation. All patients with suspected GI-GVHD underwent endoscopy and biopsy, if not contraindicated. Patients diagnosed with or suspected of having GI-GVHD were treated with steroids as per the clinical team. The patient's stool was collected on day 1 of diarrhea and post-treatment (day 3-7 on steroid if suspected or diagnosed as GI-GVHD). All included patients were followed up until 100 days after transplantation (even after discharge) or death, whichever occurred earlier.
Diagnosis of GI-GVHD (differentiating from other diarrhea etiology) and treatment protocol
GI-GVHD was diagnosed by a combination of clinical diagnosis, sigmoidoscopy (or proctoscopy), and rectal biopsy. GI-GVHD is primarily a clinical diagnosis (including histopathology that is only complementary) after excluding other causes10. As mentioned above, other causes of infective diarrhea were ruled out using microbiological (bacterial/parasitological) stool evaluation. The diagnosis of CMV colitis causing diarrhea was made if there was histopathological evidence of CMV (inclusion bodies/ immunohistochemistry staining) or rectal tissue CMV PCR detection. CMV viral syndrome (causing diarrhea) was diagnosed if the patient had fever, malaise, leucopenia, diarrhea (usually along with other organ involvement also) along with quantitative PCR detection in peripheral blood. The diagnosis of mucositis was made clinically. GI-GVHD clinical diagnosis and staging were based on the daily stool output per day11. Grades I and II were considered mild GVHD, and grades III and IV were considered severe GVHD. The histopathological grading of GI-GVHD was performed according to the method described by Shulman et al.12. All patients with a clinical suspicion of acute GVHD were started on intravenous methylprednisolone (1-2 mg/kg/day) along with optimized doses of calcineurin inhibitors. Patients with steroid-refractory GVHD received treatment with ruxolitinib, etanercept, or basiliximab.
Testing for fecal calprotectin
The collected stool samples were transported to the laboratory within 2 hours of collection and stored at
Statistical analysis
The sample size was calculated for the primary objective to assess whether FC on day 1 of diarrhea can differentiate GI-GVHD from other causes of diarrhea. We needed 50 (25 in each group) patients accounting for 80% power and 95% desired confidence level, based on a previous study by Lorenz et al.7.
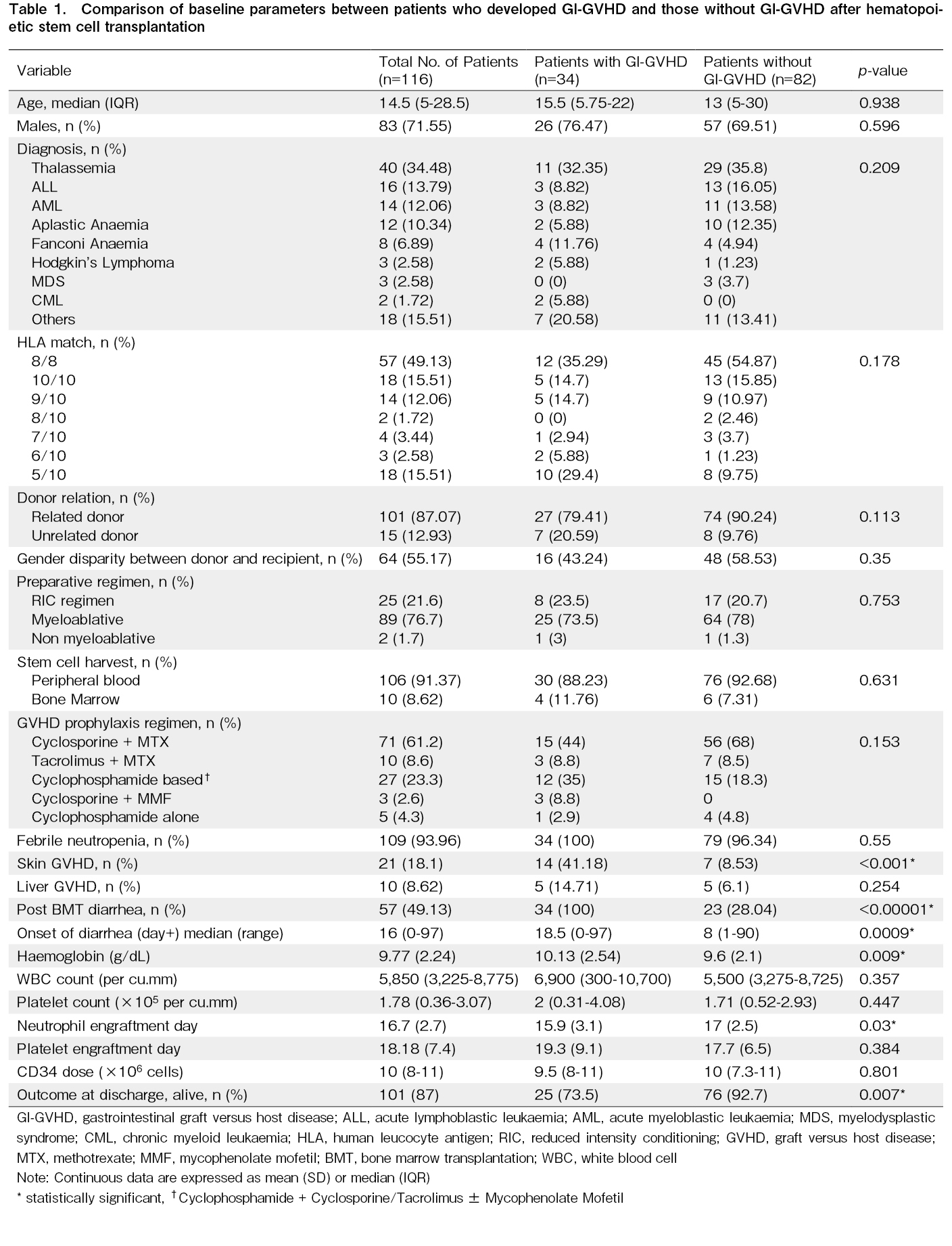
Baseline characteristics of patients in these two groups were described using standard statistical methods, with frequencies and percentages for categorical variables, means and standard deviations for continuous variables when data were not skewed, and median and quartile 1 to quartile 3 (Q1-Q3) for continuous variables when data were skewed. The chi-square test was used to examine differences in categorical variables between groups. The differences between continuous variables were analyzed using either an independent Student's t-test (non-skewed data), Mann Whitney U test (for two groups), or Kruskal Wallis test (for more than two group comparisons) (skewed data). A paired t-test was used to measure the difference in the trend of pretransplant FC to post-transplant FC. An receiver operating characteristics curve (ROC) curve analysis was performed to evaluate the ability of calprotectin to differentiate between GI-GVHD and infective diarrhea/other causes, and diagnostic accuracy measures were calculated for the optimal cut-off value that minimized false negatives. All tests were two-sided at a significance level of α=0.05. The data were analyzed using SPSS v.29 (Statistical Package for the Social Sciences, SPSS Inc., Chicago, IL, USA) and graphs were created using GraphPad Prism v.9. (GraphPad Software, San Diego, California, USA).
Results
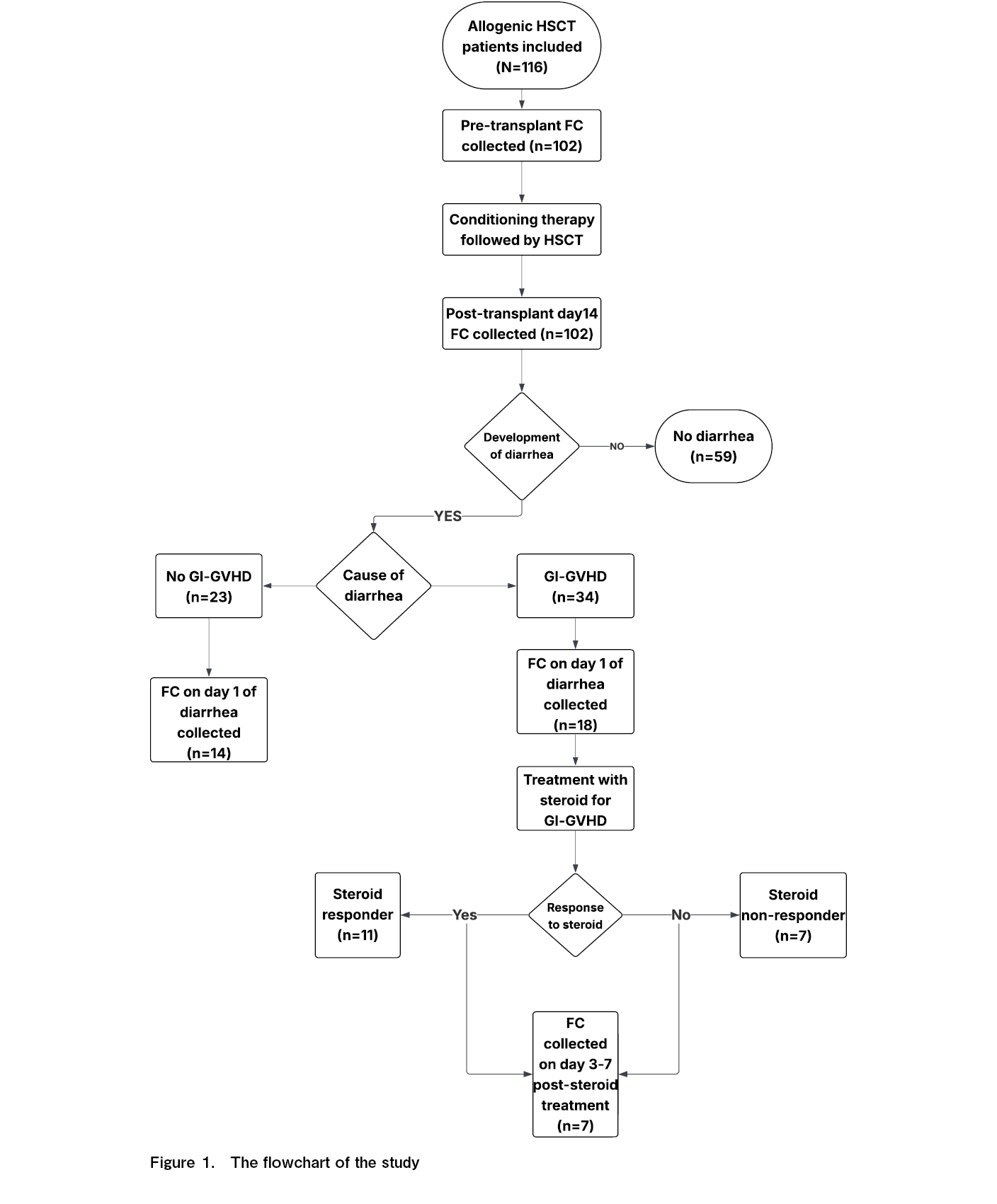
A flowchart of the study is presented in Figure 1. Of the 116 patients included in this study, 34 (29.3%) developed GI-GVHD. The baseline characteristics and comparisons between the GI-GVHD and non-GI-GVHD groups are summarized in Table 1. The median age of the patients was 14.5 years (Q1-Q3:5-28.5), and 71.6% (83/116) were male. The most common indication for HSCT was thalassemia (34.5%), followed by acute lymphoblastic leukemia (ALL) (13.8%) and acute myeloid leukemia (AML) (12%). Most patients (87.1%) received transplants from related donors.
There was no difference in age distribution of diagnosis between the patients who developed GI-GVHD and others. There was a trend toward a higher proportion of unrelated donors in the GI-GVHD group than in the other group (20.6% vs. 9.8%, p=0.11). The overall HLA-matching differences were not statistically significant between the two groups (p=0.178). However, notable differences were observed between the groups, with a larger number of HLA mismatches in the GI-GVHD group than in the non-GI-GVHD group. Most patients received myeloablative conditioning regimens, and there was no significant difference between the GI-GVHD and non-GI-GVHD groups (p=0.75). The graft source was predominantly peripheral blood (91.4%), with no significant difference between groups (p=0.631). For GVHD prophylaxis, cyclosporine and methotrexate regimens were most commonly used (61.2%), although they were less frequent in the GI-GVHD group (44% vs. 68%), followed by post-transplant cyclophosphamide (PTCy), which was more common in the GI-GVHD group (35% vs. 18.3%). The overall difference in prophylaxis was not statistically significant (p=0.153). The occurrence of skin GVHD was significantly higher in the GI-GVHD group than in the other groups (41.2% vs. 8.5%, p < 0.001). However, the occurrence of liver GVHD did not differ significantly between the two groups (14.7% vs. 6.1%, p=0.25). Patients who developed GI-GVHD had lower survival at discharge (73.5%) than those who did not develop GI-GVHD (92.7%).
Clinical presentation and treatment response in patients with GI-GVHD
Clinical GI-GVHD grading
Grade 1 GI-GVHD was observed in 11 patients (32.3%), grade 2 in eight patients (23.5%), grade 3 in four patients (11.7%), and grade 4 in 11 patients (32.3%). Diarrhea occurred in all patients with GI-GVHD. Abdominal pain affected approximately one-third of the patients (n=11), while vomiting was less common, present in only three patients.
Histopathological grading of GI-GVHD
Histopathological examination revealed grade 1 findings in 21 patients (61.8%), grade 2 histopathology in eight patients (23.5%), grade 3 in three patients (8.8%), and grade 4 in two patients.
CMV infection causing diarrhea
CMV testing revealed PCR positivity in blood samples from 7 patients (20.6%) and in tissue samples from 5 patients (14.7%). CMV was detected on histopathological examination in only one patient.
Steroid response
Twenty patients (58.8%) responded to steroid treatment, while 14 (41.2%) required second- and third-line treatment. The overall treatment outcome showed improvement with first-line therapy in 58.8% (20 patients), improvement with second-line therapy in 11.7% (4 patients), and improvement with third-line therapy in 5.9% (2 patients) of patients. Eight patients (23.5%) died despite the treatment.
Cause of non-GI-GVHD diarrhea
The causes of diarrhea in the non-GI-GVHD group included infections in four patients (17.3%: Salmonella, Shigella, C. difficile, and Aeromonas, one each), GI mucositis in two patients (8.6%), and in the other 17 patients (73.9%) no cause was identified (diarrhea was self-limiting). Five of the 17 patients underwent rectal biopsy and GVHD was ruled out. Steroids were administered to three patients: two with co-existent skin GVHD, and one with suspected GI-GVHD in whom steroids were discontinued after GVHD was ruled out on rectal biopsy. There were no other contributory conclusive diagnosis made on biopsy for the other patients.
FC in GI-GVHD diarrhea vs other causes of diarrhea
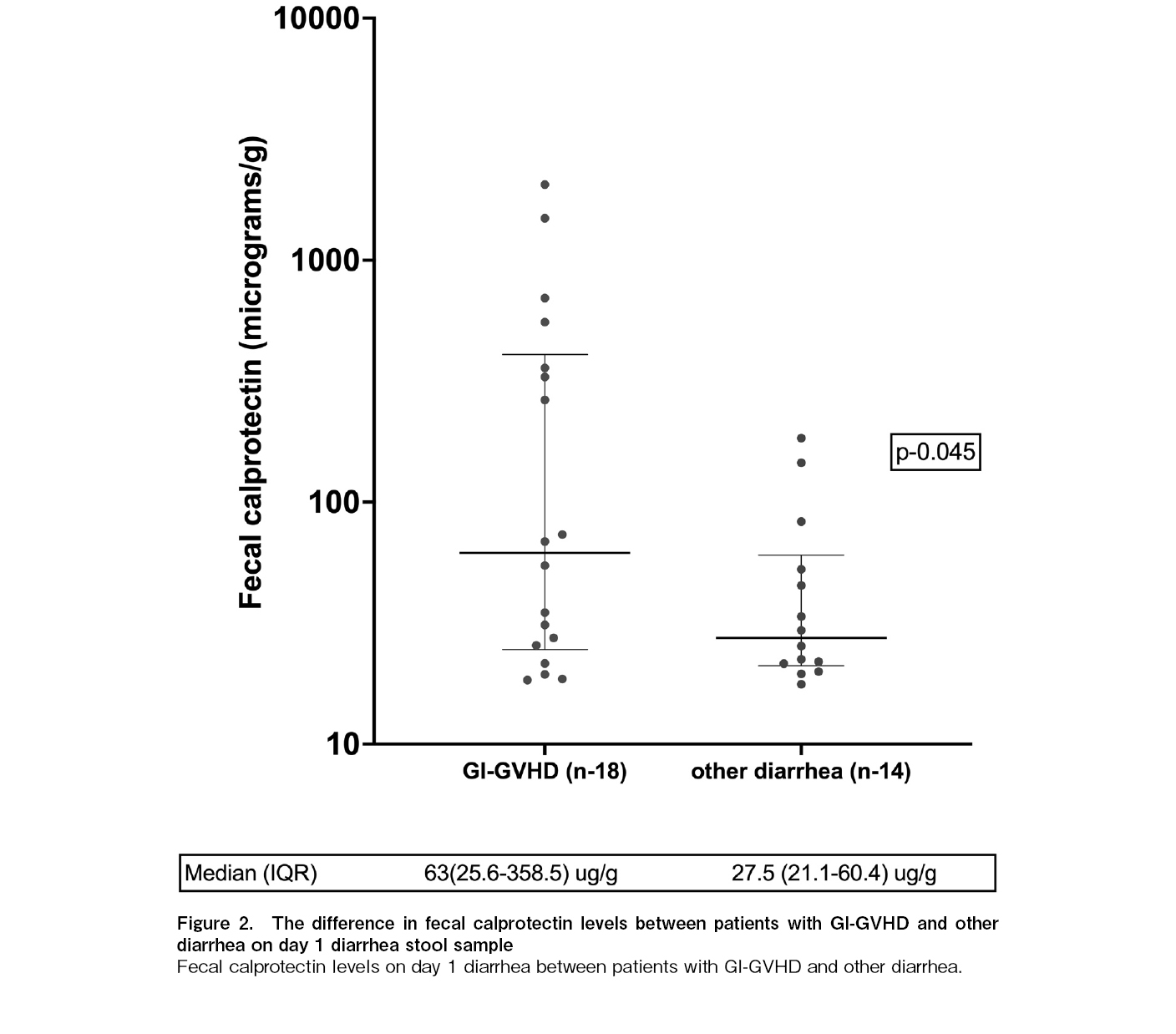
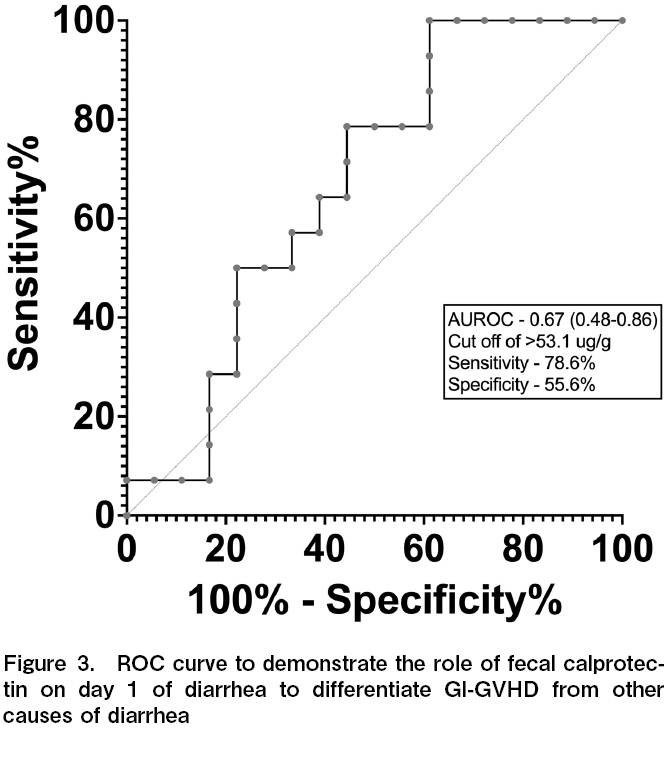
Patients with GVHD-related diarrhea demonstrated significantly higher FC levels on the day of diarrhea than those with other causes (63 [25.6-358.5] vs. 27.5 [21.1-60.4], p=0.045) (Figure 2). The cut-off value of FC for GI-GVHD diarrhea was found to be 53.1 μg/g, area under the receiver operating characteristic curve (AUROC) was 0.67 (0.48-0.86) with a sensitivity of 78.6% and specificity of 55.6% (Figure 3).
The onset of diarrhea also differed significantly between the GI-GVHD and non-GVHD cases (median day 18.5 vs. day 8, p < 0.001). Patients with GI-GVHD had a median absolute neutrophil count (ANC) of 2,500 (1,342-3,619) cells/μL on the first day of diarrhea, while those with diarrhea from other causes showed a median ANC of 5,720 (250-7,782) cells/μL (p=0.61).
Association of FC with clinical severity of GI-GVHD
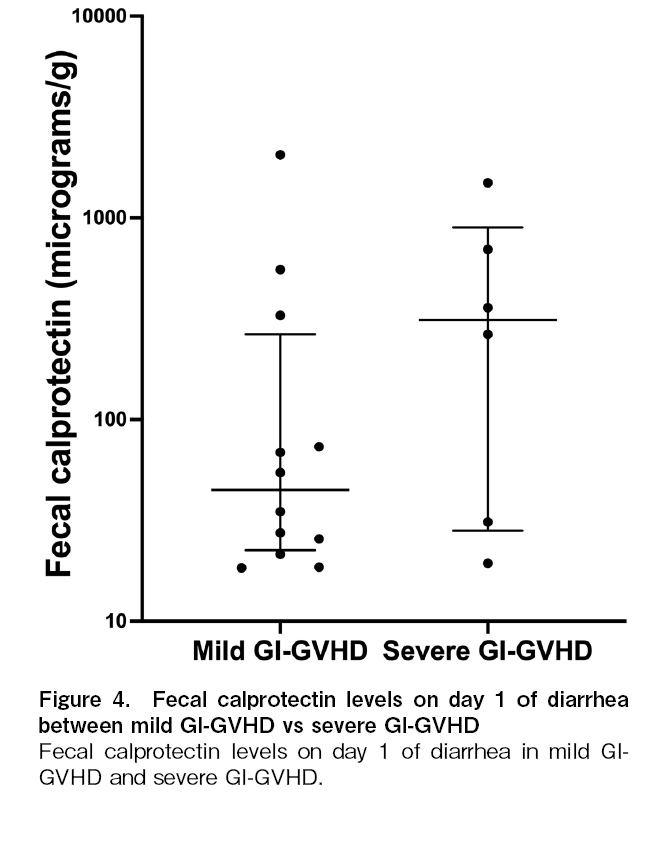
Median FC levels on the day of diarrhea in patients with severe GI-GVHD (n=6) was 311.51 (28.13-895.44) μg/g while those with mild GI-GVHD (n=12) were 44.81 (22.53-264.97) μg/g, p=0.28, Figure 4).
The FC values were not statistically different between the histopathological grades of GI-GVHD (p=0.18). Grade 1 in 21 patients (FC=61 [28.3-312.7] μg/g), grade 2 in eight patients (FC=23.6 [19.3-422] μg/g), grade 3 in three patients (FC= 527.9 μg/g), and grade 4 in two patients (FC not done in view of blood in stools).
Fecal calprotectin and steroid response
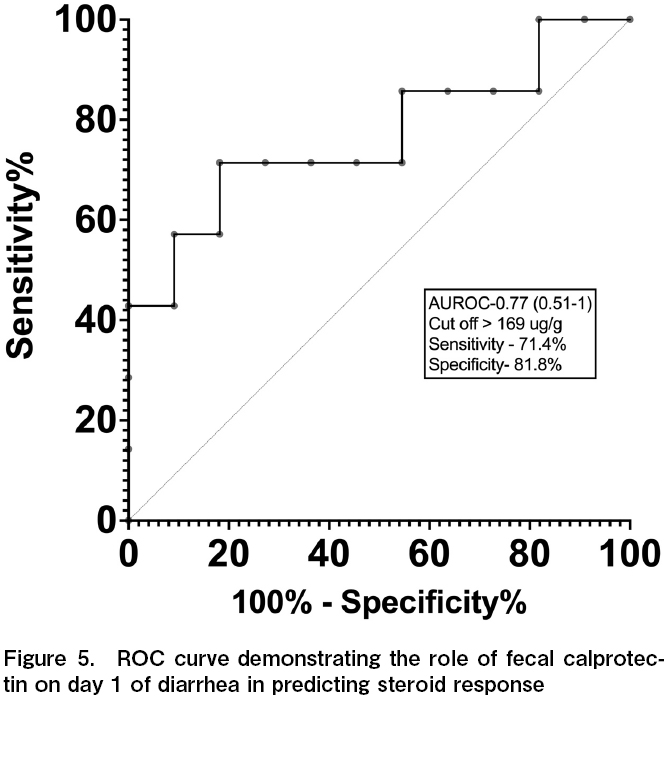
FC levels were lower for steroid responders, with the median FC being 31.2 (20.78-137.24) μg/g in steroid responders (n=11) and 311.51 (40.48-1,291.47) μg/g in steroid non-responders (n=7), p=0.034. The cut-off value of FC for steroid nonresponse was 169 μg/g; AUROC was 0.77 (0.51-1.0) with a sensitivity of 71.4% and specificity of 81.8% (Figure 5).
Among patients with GI-GVHD, 58.8% improved with first-line therapy (steroids), 12.5% with second-line therapy, and 6.2% with third-line therapy. The mortality rate of the patients with GVHD was 25%.
Predictors of steroid response among GI-GVHD
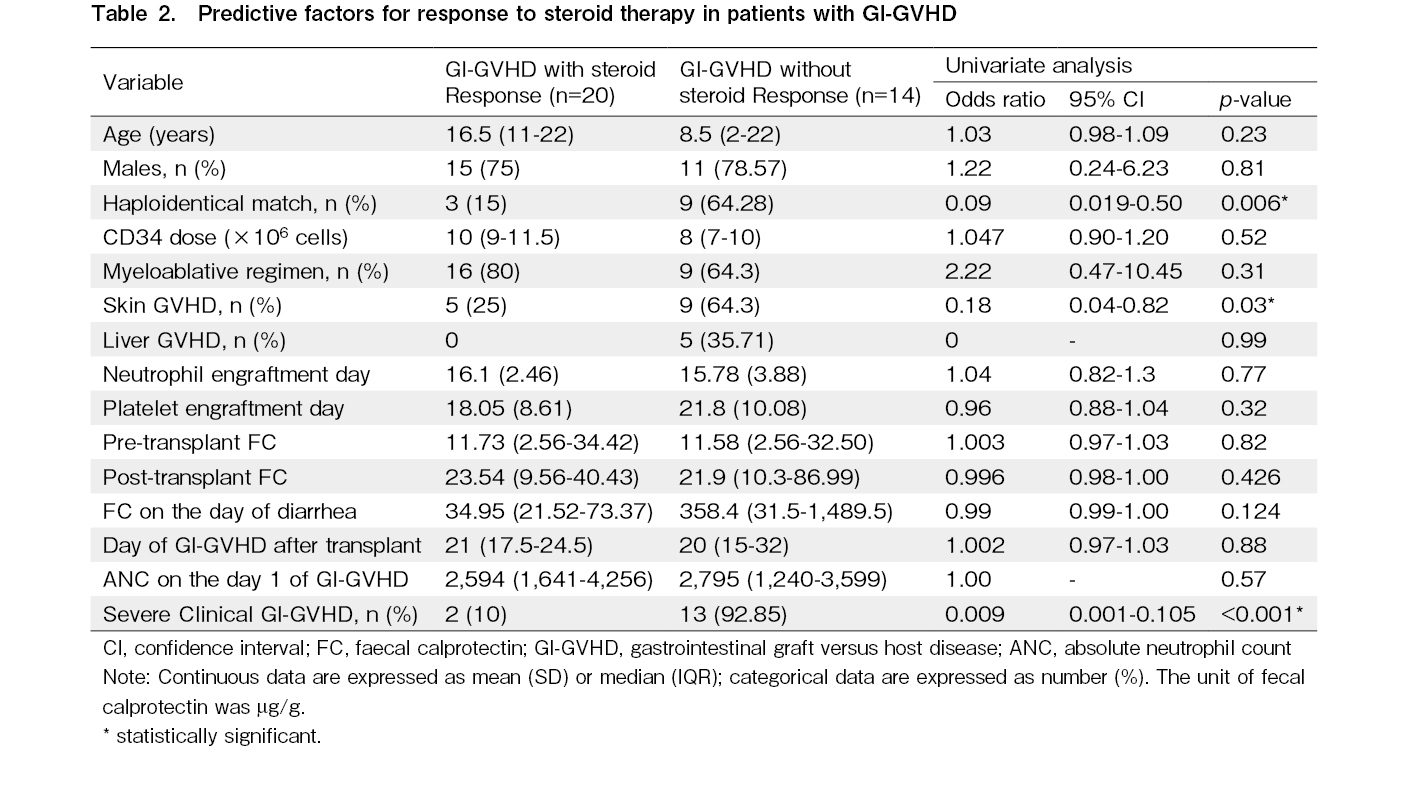
Predictors of response to steroids were studied in all GI-GVHD patients (Table 2). On univariate analysis, haploidentical match (odds ratio (OR): 0.09, 95% confidence interval (CI): 0.019-0.5, p=0.006), co-existent skin GVHD (OR: 0.18, 95% CI: 0.04-0.82, p=0.03), and severe clinical GI-GVHD (OR: 0.009, 95% CI: 0.001-0.105, p<0.001) were found to be significant predictors for steroid non-response in GI-GVHD. Multivariate analysis was not possible due to significant co-linearity between these variables that were significant in univariate analysis.
On-treatment fecal calprotectin among GI-GVHD patients (responder vs non-responder)
This time-point sampling was performed for only 18 patients. There was no significant difference in on treatment (steroid) day 3-7 FC levels between steroid responders (n=10) and steroid non-responders (n=8) after 3-7 days of steroid treatment for GI-GVHD (28.32 vs 52.77, p=0.93).
Prognostic value of pre-diarrhea fecal calprotectin
Pre-transplant FC levels were similar between patients who later developed GI-GVHD and those who did not (12.12 [10.64-36.29] vs 16.32 [9.5-51.28] μg/g, p=0.9681). Early post-transplant FC levels (day 14) also did not differ between GI-GVHD and other causes of diarrhea (26.02 [10.04-47.09] vs. 11.01 [10.14-33.54] μg/g, p=0.14). Median FC levels in GI-GVHD patients showed an upward trend from pre-transplant (12.12 [10.64-36.29] μg/g) to post-transplant (26.02 [10.04-47.09] μg/g), though this increase was not statistically significant (p=0.17). Conversely, patients with diarrhea from other causes experienced a reduction in median FC levels from pre-transplant (16.32 [9.5-51.28] μg/g) to post-transplant (11.01 [10.14-33.54] μg/g, p=0.72).
Discussion
Our study demonstrated the potential utility of FC as a biomarker for GI-GVHD diagnosis, severity assessment, and prediction of treatment response.
The significantly higher FC levels in GVHD-related diarrhea than in non-GVHD diarrhea (62.97 vs 27.46 μg/g, p=0.045) supports its role as a diagnostic biomarker. Our finding aligns with that of a previous study by Rodriguez et al., however, we suggest FC cut off value of 53.1 μg/g for a higher sensitivity and lower specificity compared to Rodriguez et al. who suggested a cut off value of FC 100 μg/g with sensitivity of 31% but specificity of 90%6. Prior study by Lorenz et al., had shown much FC value 850 μg/g vs 119 μg/g (GI-GVHD vs rest)7. The later onset of GVHD diarrhea (median day 18.5) compared to infectious diarrhea (median day 8) provides an additional temporal parameter for differential diagnosis. O'Meara et al.8 similarly reported that the timing of diarrhea onset can help differentiate GVHD diarrhea from other causes.
FC levels showed significant predictive value for steroid response, with non-responders having nearly ten-fold higher levels compared to responders (311.51 vs 31.2 μg/g, p=0.034). This aligns with the findings of Broglie et al.13 and O'Meara et al.8, who demonstrated that higher FC levels correlate with poor treatment response. Both Broglie et al.13 and O'Meara et al.8 have shown that the FC values of steroid non-responders were well above 400 μg/g, we found values just above 300 μg/g for steroid non responders.
While studying the relation between clinical grade of GI-GVHD and FC values, we found severe (grade 3 and grade 4) GI-GVHD compared to mild GI-GVHD (grade 1 and grade 2) had significantly higher FC values. However, the histopathological grades did not show a serial increase in FC with grade 2 had lower FC than grade 1 of GI-GVHD and grade 3 had higher FC values, we did not do FC for grade 4 (in view of blood in stools). This discrepancy of FC values lower in grade 2 than grade 1 maybe secondary had very few number of patients in grade 2 GI-GVHD and this may also have led to a skewed results. It is also to be noted that clinical grade of GI-GVHD is a better predictor of outcome and the correlation between clinical and histopathological grades of GI-GVHD are modest.
In our study, we observed no significant difference in the ANC on the first day of diarrhea between patients with GI-GVHD and those with diarrhea of other etiologies. While neutropenia is a recognized risk factor for various post-transplant complications, our results indicate that both GI-GVHD and non-GVHD diarrhea can occur across a wide spectrum of neutrophil counts.
Our results show that there is no role for pre-emptive testing of FC pre- and post-transplantation. We excluded patients with frank blood in stools because blood interferes with accurate FC estimation (resulting in falsely elevated values). As a consequence of this exclusion criterion, there were many missing samples from patients with severe GI-GVHD.
Fecal alpha-1-antitrypsin has also been shown to have a good AUROC for diagnosis of GI-GVHD; however, the use of this marker has not translated into clinical practice6. MicroRNA (miRNA-155, 146a) has also been suggested as a marker for diagnosis and a potential target for treatment in acute GI-GVHD; this needs more validation and human studies14,15.
Our study further validates the use of FC in the diagnosis of GI-GVHD as shown in another study from India which showed similar results, however, the cut off suggested in the study by Puntar et al. for FC was 100 μg/g, however the authors of the study aimed for higher specificity and but we aimed for higher sensitivity16. We embarked on the study to establish a cut-off which is relevant for Indian population and we found our FC values in GI-GVHD diarrhea and steroid non-response GI-GVHD were similar to that reported by studies from other parts of the world. However, we aimed to have a higher sensitivity thereby we suggest a FC cut-off of 53.1 μg/g, compared to prior studies which have suggested values of 100 μg/g. The strengths of this study include the comprehensive assessment of FC at multiple time points, correlation with clinical and histopathological grading, and evaluation of FC as a predictor of steroid response. The real-time results of FC can guide bedside treatment strategies to respond early to steroid-refractory GVHD, a complication associated with significant morbidity and mortality.
The limitations of this study include its single-center design, missed sample collection (primarily due to the presence of blood in stools) in a few patients and we did not perform rectal biopsy for all patients with diarrhea (if done would have made the diagnosis of GI-GVHD vs other etiologies more robust). Larger studies are needed, particularly assessing FC levels at the onset of diarrhea in patients with GI-GVHD, to derive an accurate cut-off to predict steroid responsiveness. Studies incorporating FC monitoring during GI-GVHD treatment will further help evaluate FC as a serial bedside severity assessment tool, along with clinical response, to improve decision-making in the management of GI-GVHD.
To conclude, a higher FC value (> 53.7 μg/g) on day 1 of diarrhea could be a marker to aid in the diagnosis acute GI-GVHD. Higher FC levels (> 169 μg/g) could predict a poor response to steroid treatment.
Author Contributions
SEK: Conceptualization, software, validation, formal analysis, investigation, data curation, original draft preparation, and final approval of the manuscript, SS: Conceptualization, validation, data curation, review and editing, and final approval of the manuscript, TV: Investigation, data curation, review and editing, and final approval of the manuscript, PKB: Investigation, data curation, review and editing, and final approval of the manuscript, KSL: Investigation, data curation, review and editing, and final approval of the manuscript, SH: Investigation, data curation, review and editing, and final approval of the manuscript, JAJ: Conceptualization, methodology, review and editing, and final approval of the manuscript, AKD: Conceptualization, methodology, review and editing, and final approval of the manuscript, VM: Conceptualization, methodology, validation, project administration, formal analysis, data curation, review and editing, and final approval of the manuscript, BG: Conceptualization, methodology, validation, project administration, formal analysis, data curation, review and editing, and final approval of the manuscript
Conflicts of Interest
The authors declare no conflict of interest. Disclosure forms provided by the authors are available on the website.
Funding Statement
This work was supported by Christian Medical College, Vellore Research Office Internal-fluid research grant (IRB min. No. 10394 dated 3rd Nov 2016)
Data Availability Statement
The data set generated during and/or analysed during the current study is available from the corresponding author at a reasonable request.
References
1.Uberti JP, Deol A, Ratanatharathorn V. Pathophysiology, prevention, and treatment of acute graft-versus-host disease. Transpl Res Risk Manag. 2011; 3: 31-44.
2.Sung AD, Chao NJ. Concise Review: Acute Graft-Versus-Host Disease: Immunobiology, Prevention, and Treatment. Stem Cells Transl Med. 2013; 2: 25-32.
3.McDonald GB. How I treat acute graft-versus-host disease of the gastrointestinal tract and the liver. Blood. 2016; 127: 1544-50.
4.Malik MN, Rafae A, Durer C, Durer S, Anwer F. Fecal Calprotectin as a Diagnostic and Prognostic Biomarker for Gastrointestinal Graft Versus Host Disease: A Systematic Review of Literature. Cureus. 2019; 11: e4143.
5.Kumar SE, Jaleel R, Varghese T, Praharaj I, Benny S, Chowdhury SD, et al. Fecal calgranulin as a potential screening tool to differentiate inflammatory bowel disease from irritable bowel syndrome. Indian J Gastroenterol. 2024; 43: 264-6.
6.Rodriguez-Otero P, Porcher R, de Latour RP, Contreras M, Bouhnik Y, Xhaard A, et al. Fecal calprotectin and alpha-1 antitrypsin predict severity and response to corticosteroids in gastrointestinal graft-versus-host disease. Blood. 2012; 119: 5909-17.
7.Lorenz F, Marklund S, Werner M, Palmqvist R, Wahlin BE, Wahlin A. Fecal calprotectin as a biomarker of intestinal graft versus host disease after allogeneic hematopoietic stem cell transplantation. Sci Rep. 2015; 5: 7920.
8.O'Meara A, Kapel N, Xhaard A, Sicre de Fontbrune F, Manéné D, Dhedin N, et al. Fecal calprotectin and α1-antitrypsin dynamics in gastrointestinal GvHD. Bone Marrow Transplant. 2015; 50: 1105-9.
9.Praharaj I, Revathy R, Bandyopadhyay R, Benny B, Azharuddin Ko M, Liu J, et al. Enteropathogens and Gut Inflammation in Asymptomatic Infants and Children in Different Environments in Southern India. Am J Trop Med Hyg. 2018; 98: 576-80.
10.Iqbal N, Salzman D, Lazenby AJ, Wilcox CM. Diagnosis of gastrointestinal graft-versus-host disease. Am J Gastroenterol. 2000; 95: 3034-8.
11.Tuncer HH, Rana N, Milani C, Darko A, Al-Homsi SA. Gastrointestinal and hepatic complications of hematopoietic stem cell transplantation. World J Gastroenterol. 2012; 18: 1851-60.
12.Shulman HM, Kleiner D, Lee SJ, Morton T, Pavletic SZ, Farmer E, et al. Histopathologic diagnosis of chronic graft-versus-host disease: National Institutes of Health Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease: II. Pathology Working Group Report. Biol Blood Marrow Transplant. 2006; 12: 31-47.
13.Broglie L, Rademaker A, Galvin J, Ray A, Tse WT, Duerst R, et al. Fecal calprotectin and serum albumin as markers of gastrointestinal graft versus host disease. Hematol Oncol Stem Cell Ther. 2018; 11: 169-74.
14.Ranganathan P, Heaphy CEA, Costinean S, Stauffer N, Na C, Hamadani M, et al. Regulation of acute graft-versus-host disease by microRNA-155. Blood. 2012; 119: 4786-97.
15.Pitea M, Canale FA, Porto G, Verduci C, Utano G, Policastro G, et al. The Role of MicroRNA in Graft-Versus-Host-Disease: A Review. Genes. 2023; 14: 1796.
16.Puntar S, Nayak L, Mantri N, Ramadwar M, Patil P, Mehta S, et al. Diagnostic Value, Predictive Significance and Kinetics of Fecal Calprotectin in Suspected Acute Gut Gvhd / Gvhd Flare Post Allogeneic Stem Cell Transplant. Blood. 2024; 144 (Suppl 1): 4901.
Search
Actions
News