Volume 9 (2026) Issue 1 No.1 Pages 1-8
Abstract
Background: There is paucity of data on the outcomes of allogeneic stem cell transplantation (allo-SCT) for myelofibrosis, from developing countries.
Materials and Methods: This is a retrospective analysis of patients undergoing allogeneic stem cell transplant for myelofibrosis at our center between January 1998 to December 2023.
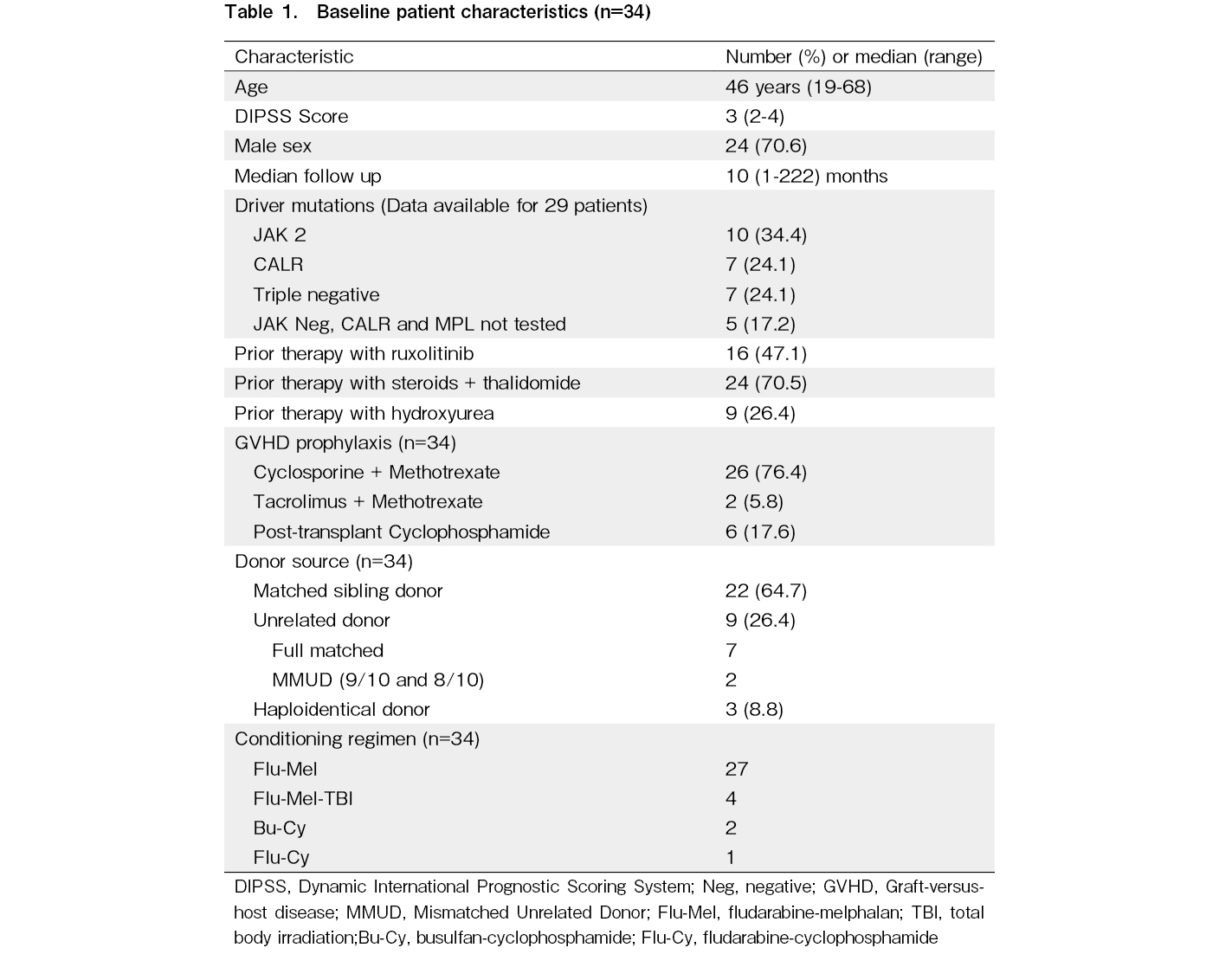
Results: Thirty-four patients underwent allogeneic transplantation with a median age of 46 (19-68) years with 24 (70.6%) being males. The median dynamic international prognostic scoring system (DIPSS) Score was 3 (2-4). JAK2 was the most common driver mutation, in 10 (34.5%) patients. Majority of patients received reduced intensity conditioning with Fludarabine-Melphalan +/-total body irradiation (n=32) and all had peripheral blood grafts with donor source being matched sibling in 22 (64.7%), unrelated donor in 9 (26.5%) [7 full HLA match, one 9/10 HLA match and one 8/10 HLA match] and haploidentical in 3 (8.8%). The median CD34 cell dose infused was 9.9 (3.4-22) × 106/kg. The majority received calcineurin inhibitor + methotrexate (n=28) as Graft versus Host Disease (GVHD) prophylaxis. Two patients died before day 14 due to sepsis while 3 (9.3%) had primary graft failure. Neutrophil engraftment occurred in 29 patients (85.3%) at a median of 15 (range 8-25) days. Acute GVHD was noted in 20 (58.8%) patients with grade 3-4 GVHD in 13 (38.2%). Chronic GVHD was noted in 12 patients with 4 being extensive chronic GVHD as per revised Seattle criteria. On last follow up, 15 (44.1%) patients are alive while 19 have died.
Conclusion: Allo-SCT for myelofibrosis is associated with reasonable cure rates, however strategies to reduce graft failure and GVHD are required.
Introduction
Myelofibrosis is clonal disorder, characterized by bone marrow fibrosis and inefficient hematopoiesis leading to worsening cytopenia, splenomegaly and constitutional symptoms. It has the worst prognosis amongst all the myeloproliferative neoplasms1. Despite newer advances in treatment, stem cell transplantation (SCT) remains the only curative option2. Though newer targeted molecules like JAK inhibitors are being used, its role in improving overall survival is still limited. SCT in patients with myelofibrosis is challenging and is associated with poorer outcomes compared to other hematological malignancies2. Various studies have showed long term relapse free survival of 40-60%3. There are many pre and post-transplant clinical and laboratory parameters that are known to influence outcomes2,4,5, and the risk of transplant related mortality versus the risk of disease related mortality determines the timing of transplant5. Over the years, various risk stratification tools and scoring systems have been developed and are recommended for an individualized treatment selection6. Engraftment failure and post-transplant graft versus host disease (GVHD) are predominant problems associated with HSCT for myelofibrosis7. With better targeted therapies being identified for myelofibrosis, the decision about when and whom to transplant has still not been solved, but has rather become more complex8. Because of the rarity of the disease and low number of patients undergoing transplant, there is limited data on myelofibrosis transplants, especially from India.
In this study we performed a retrospective analysis of pre-transplant clinical, laboratory parameters and outcomes in patients with myelofibrosis who underwent their first allogeneic bone marrow transplantation at our center.
Materials and Methods
This is a retrospective single-center analysis that included patients diagnosed to have primary myelofibrosis and underwent their first allogeneic bone marrow transplantation at Christian Medical College and Hospital, Vellore, India between June 1998 to December 2023. All the data was retrieved from Hospital Electronic Medical Records and hospital databases. This study was approved by the local Institutional Review Board, ref. IRB Min No. 14818 dated 31.08.2022. Since it was a retrospective study, waiver of consent was obtained from Institutional Review Board.
Data was collected on pre-transplant clinical, laboratory parameters and outcomes in patients with myelofibrosis who underwent allogeneic SCT. Our cohort included both primary (idiopathic) myelofibrosis and secondary myelofibrosis (post-polycytemia vera [PRV] and post-essential thrombocythemia [ET]). Parameters studied included dynamic international prognostic scoring system (DIPSS) score (consists of age > 65 years, hemoglobin < 10 g/dL, leukocytes > 25 × 109/L, circulating blasts ≥ 1% and constitutional symptoms), co-morbidities, Karnofsky score, splenectomy status prior to SCT, cytogenetics, and molecular markers if available, type of HLA match, conditioning regimen and type of graft used4.
The conditioning regimen used was based mainly on performance status of the patient and associated comorbidities. Conditioning regimens used included myeloablative transplants such as Busulfan + Cyclophosphamide (Bu-Cy) or reduced intensity conditioning (RIC) transplants such as fludarabine with melphalan (Flu-Mel) or fludarabine with cyclophosphamide (Flu-Cy). Total body irradiation (TBI) 200 cGy was included in the conditioning for patients undergoing a haploidentical stem cell transplant. Anti-thymocyte globulin was not routinely used except in three unrelated donor transplants. Donors included matched sibling (MSD), unrelated donors (seven full HLA matched, one 9/10 HLA matched and one 8/10 HLA matched) and haplo-identical (Haplo) donors. Supportive care was provided as per institutional practice. All patients received antibacterial prophylaxis with Penicillin G, antiviral prophylaxis with acyclovir, and antifungal prophylaxis with fluconazole (oral/IV as tolerated). Cotrimoxazole was administered for Pneumocystis jirovecii prophylaxis after engraftment. Transfusion support was given to maintain hemoglobin above 7 g/dL and platelet counts above 20 × 109/L. Infections were treated with appropriate antibiotics and antifungals as per culture reports. Pre-transplant fungal screening was performed only in symptomatic patients using chest imaging and galactomannan testing, but universal screening was not consistently implemented. Antifungal drug-level monitoring was not routinely available during the earlier years of the cohort and was not done consistently.
In patients with myelofibrosis, higher CD34+ cell doses were infused when available, with the aim of promoting engraftment in the setting of marrow fibrosis and splenomegaly.
Neutrophil and platelet engraftment was identified as absolute neutrophil counts > 0.5 × 109/L for 3 consecutive days and platelet count > 20 × 109/L for 7 consecutive days without transfusion9,10. Donor chimerism was evaluated using Variable Number Tandem Repeat (VNTR) analysis on days +30, +60, +90 and when clinically indicated otherwise.
Transplant outcomes were described in terms of overall survival, acute and chronic GVHD, infections and transplant related mortality (TRM). The time from SCT until death from any cause was defined as the overall survival (OS). Modified Glucksberg and Modified Seattle criteria were used to grade acute and chronic GVHD11,12.
Statistical Analysis
Descriptive statistics were used, such as mean (standard deviation) / median (range) for clinical and laboratory data. For categorical variables, numbers and percentages were used. Survival analysis was performed using the Kaplan Meier method. A p-value < 0.05 was regarded as statistically significant. All analysis was performed using SPSS version 21.0 (SPSS Inc, Chicago, IL).
Results
During the study period, 34 patients (24 males and 10 females) underwent first allogeneic SCT for myelofibrosis at our center with a median age of 46 (range: 19-65) years. Pre-transplant characteristics are described in Table 1. At the time of transplant, 27 patients (79.4%) were transfusion dependent and 25 of 34 patients (73.5%) had massive splenomegaly, defined as a spleen palpable > 8 cm below the left costal margin, crossing the midline, or measuring > 20 cm on imaging. The median time from diagnosis to SCT was 28 months (1-135). Majority of the patients (70.5%) received a combination of thalidomide and prednisolone prior to SCT, while 16 (47.1%) patients received ruxolitinib. Few patients (26.4%) had also received hydroxyurea as treatment prior to SCT. One patient was taken up for SCT without prior exposure to any drugs while another patient underwent splenectomy 3 years prior to SCT.
The median DIPSS Score at the time of SCT was 3 (range: 2-4). Six patients (17.6%) were classified as intermediate -1 risk and 28 (82.3%) as intermediate -2 risk, as per DIPSS score at the time of SCT. One patient had secondary myelofibrosis while rest of the 33 patients had primary myelofibrosis. Of the 24 patients where data on all 3 driver mutations was available,
Co-morbidities were present in 16 patients (47.1%) including diabetes mellitus in 11, hypothyroidism in 5, hypertension in 4, coronary artery disease in 2, tuberculosis in 1, post tuberculosis (Tb) destructed lung in 1, pulmonary thromboembolism in 1 and hepatitis B related chronic liver disease (CLD) in one patient. The median Karnofsky score was 90 (range: 70-100, n=26).
Transplantation
Majority of the patients received reduced intensity conditioning (RIC) with Flu-Mel +/-TBI (n=31) while 2 received Bu-Cy and 1 patient received
Engraftment, toxicity and GVHD
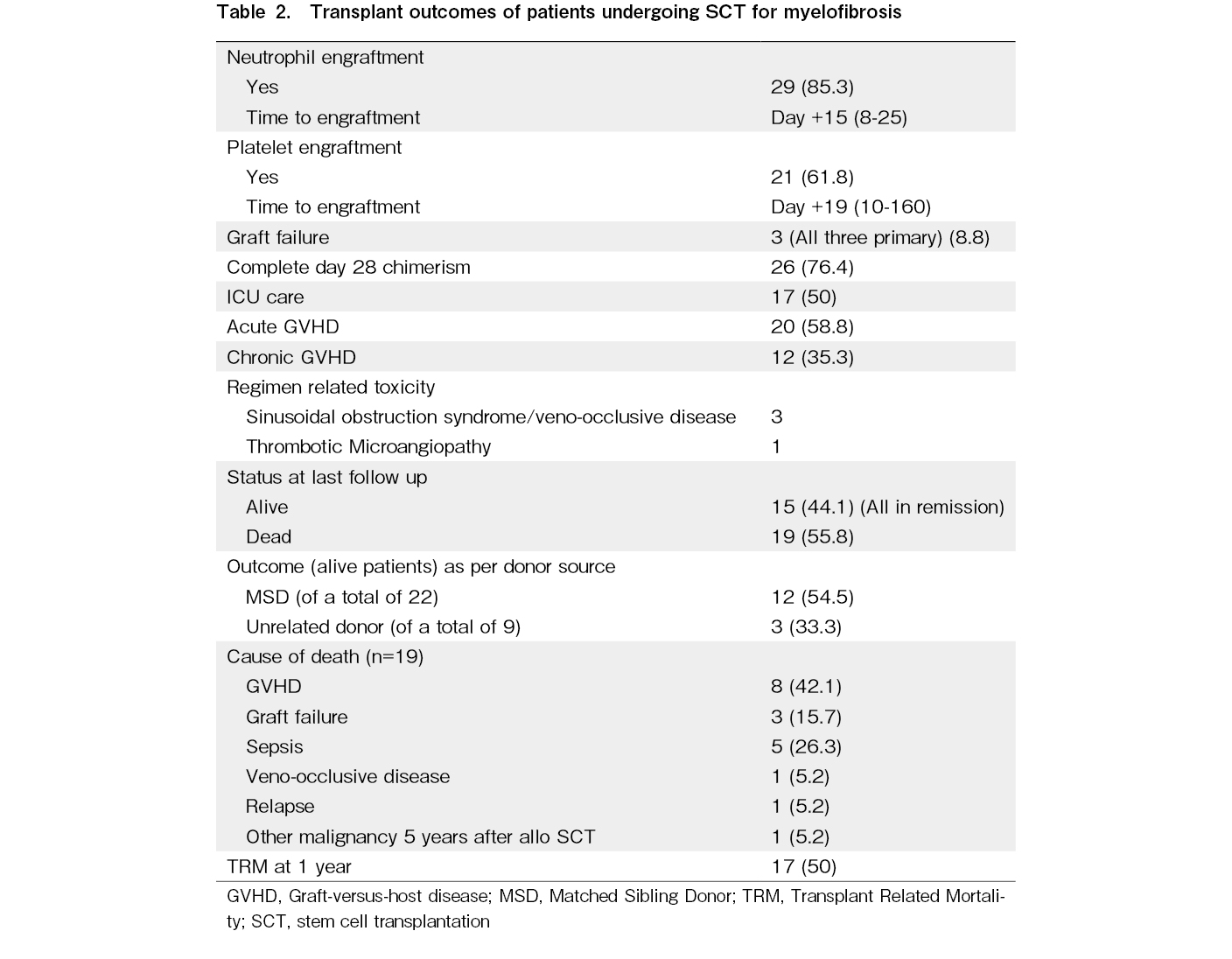
Table 2 describes the outcomes of SCT. The median infused CD34 cell dose was 9.9 (3.42-22) × 106 cells/kg. Neutrophil engraftment occurred in 29 patients (85.3%) at a median of 15 days (range: 8-25) while 2 patients died prior to day 14 due to sepsis and 3 (9.3%) had primary graft failure.
Chimerism analysis showed that at day +30, 26 patients (76.5%) had complete donor chimerism and 1 patient had mixed chimerism. At day +60, complete donor chimerism was documented in 18 patients, and at day
The cumulative incidence of acute GVHD was 58.8% with grade 3-4 GVHD in 38.2%. Chronic GVHD was noted in 12 patients with 4 being extensive chronic GVHD as per revised Seattle criteria12.
Twenty-eight patients had documented infections including blood stream bacterial infections (BSI) in 20 (58.8%), invasive fungal disease (IFD) in 13 (38.2%) and cytomegalovirus (CMV) reactivation in 12 (35.2%). Three patients had BSI with carbapenem resistant organisms (CRO) while 1 had BSI with methicillin resistant Staphylococcus aureus (MRSA). Of the 13 patients with IFD, 4 patients had isolated fungus in blood culture – one each had Fusarium dimurium, Candida auris, Candida tropicalis and one had broad aseptate fungi – Rhizopus arrhizus, candida tropicalis and fusarium in two sequential cultures and rest were either probable or possible IFD. CMV reactivation requiring treatment was documented in 12 patients while symptomatic BK viral infection occurred in 4 patients.
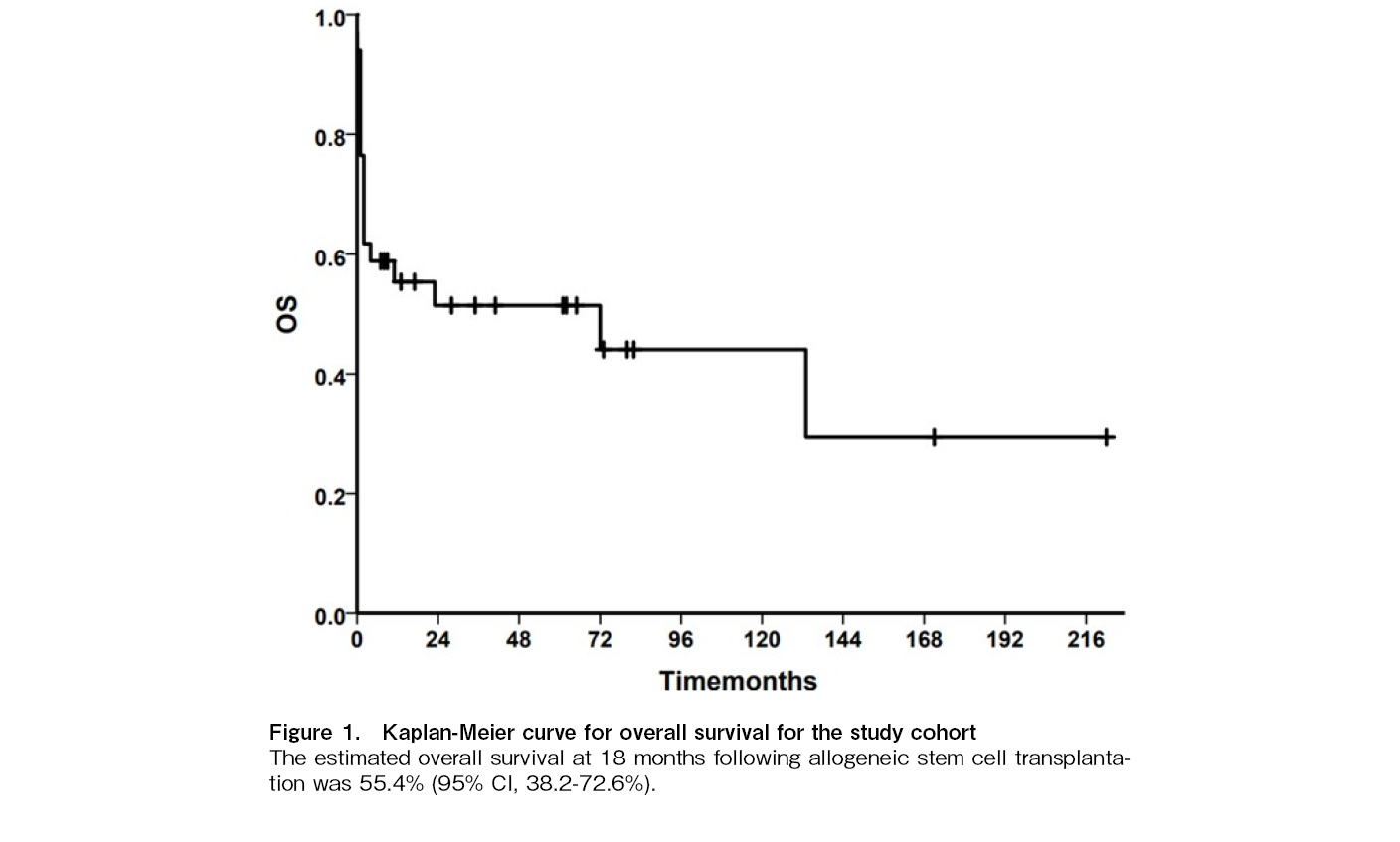
At a median follow up of 10 months (range: 1-212), 15 patients are alive (44.1%) while 19 (55.8%) have died. At 18 months following allogeneic stem cell transplantation, the OS was 55.4% (95% CI, 38.2-72.6%). The GVHD-free, relapse-free survival (GRFS) at the same time point was 47.1% (95% CI, 29.9-64.3%).
Causes of death included GVHD (n=8) followed by sepsis (n=5), graft failure (n=3), VOD (n=1) while 1 patient progressed to acute myeloid leukemia 8 years after SCT and died subsequently following a second transplant and one patient developed vertebral sarcoma after 5 years of SCT and died of pneumonia. Survival outcomes were better in patients undergoing a matched sibling donor transplant (54.5%) compared to unrelated donor transplant (33.3%) and haplo-identical transplants (all died) (Figure 1). Survival appeared higher in patients transplanted after 2010 (13/26, 50%) compared to those transplanted before 2010 (2/8, 25%), although the cohort size was too small for statistical analysis.
On univariate analysis, donor type was found to be the predictor of outcomes, with matched sibling donor transplant having significantly better outcomes than MUD or haplo SCT. Other variables such as age, sex, time from diagnosis to SCT, DIPSS score, exposure to ruxolitinib, cell dose, time of engraftment, acute and chronic GVHD did not affect outcomes.
Discussion
Though allogeneic (allo)-SCT remains the only potentially curative option for myelofibrosis, considering its complications, choosing optimal patients for transplant becomes essential5,8,13. Treatment options for myelofibrosis include androgens, steroids, thalidomide, lenalidomide and newer agents which act on JAK2 signaling pathway. Most of these drugs have limited utility in low and middle income countries (LMICs) considering their high cost and indefinite duration of treatment6. Multiple scoring systems are available for prognostication and choosing treatment options for myelofibrosis. The most commonly used score is the DIPSS which is based on clinical features and minimal laboratory parameters and thus simple to calculate4. Newer scoring systems include genetically inspired prognostic scoring system (GIPSS), mutation-enhanced international prognostic scoring system (MIPSS)-70 and MIPSS-70 + version2 which take into account the cytogenetic abnormalities, high risk mutations, new sex- and severity-adjusted hemoglobin thresholds14–16. At present, most guidelines recommend allogeneic stem cell transplantation for patients in DIPSS intermediate-2 and high risk patients5. Latest treatment update suggests approach based on MIPSS70 + version2 score, with high risk and very high risk patients are recommended to go for allo-SCT, if they are transplant eligible17. Asymptomatic low risk and very low risk patients should be offered careful observation and should be offered investigational treatment if they are symptomatic while Intermediate risk 1 patients need individualized approach17.
Studies all over the world for allogeneic stem cell transplants for myelofibrosis have shown overall outcome between 40-60% with major obstacles being engraftment failure and GVHD leading to high TRM8,18–21. Massive splenomegaly and a fibrotic bone marrow with proinflammatory stem cell niche leads to engraftment failure, delayed engraftment, and poor graft function7,22. Measures to reduce spleen size prior to SCT include the use of JAK 2 inhibitors, splenectomy or splenic irradiation, however each of these modalities comes with limited evidence. Delay in engraftment leads to higher risk of infections and sepsis, especially in LMICs. Sepsis was second most common cause of death in our study following GVHD.
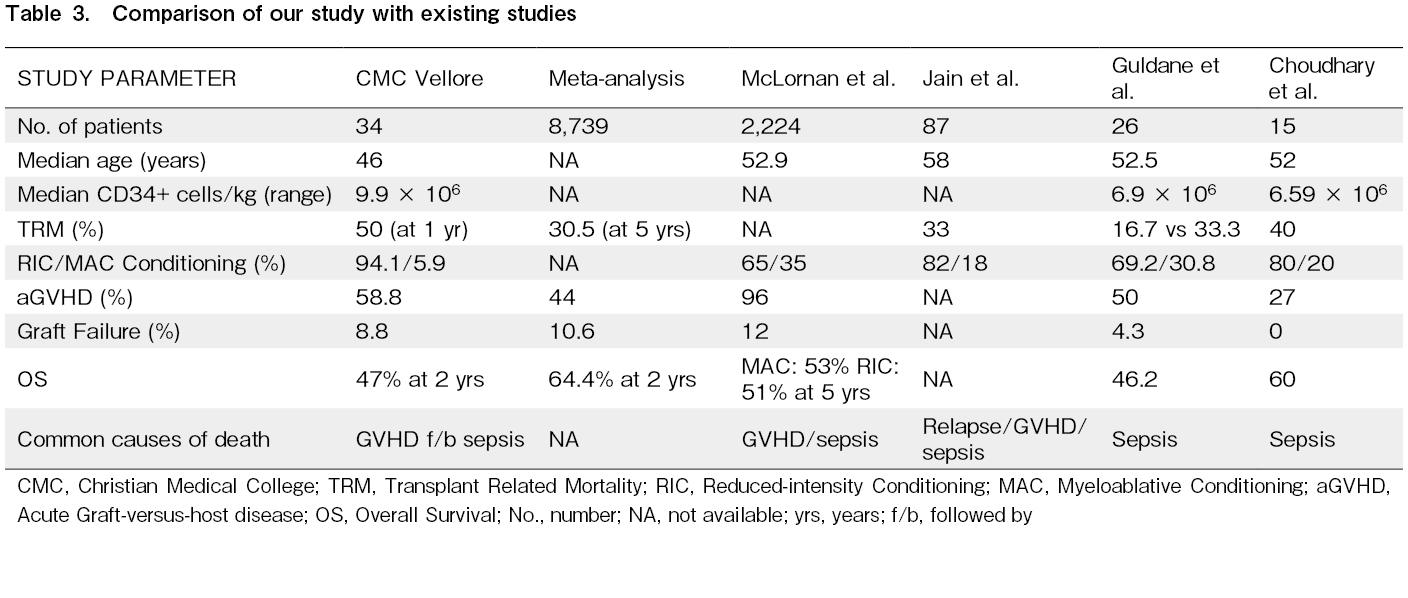
As this is a relatively uncommon disease with fewer numbers of patients going for SCT, data available for allo-SCT in myelofibrosis is scarce, especially in LMICs. Many other questions apart from optimal timing, including role of JAK inhibitors, optimal conditioning, role of splenectomy or splenic irradiation pre-transplant are still unanswered. A recent meta-analysis by Bewersdorf et al. evaluating outcomes of allo-HCT in patients with myelofibrosis, included forty-three studies with 8,739 patients3. The overall survival rates were 66.7%, 64.4% and 55% at 1, 2, and 5-years respectively with a 1 year mortality of 25.9%. The combined rate of graft failure was 10.6% with primary and secondary graft failure occurring in 7.3% and 5.9% of patients, respectively. The rates of acute and chronic GVHD were 44.0% (grades III-IV, 15.2%) and 46.5% (extensive, 26.1%), respectively3. The European Bone Marrow Transplant (EBMT) working group published a large retrospective study in 2016, including 2,916 patients, evaluating the determinants of SCT outcomes in myelofibrosis24. After a median follow-up of 4.7 years from transplant, median survival was 5.3 years with the estimated overall OS at 1, 3, and 5 years were 66%, 55%, and 50%, respectively. The rates of acute and chronic GVHD were 46% (grades III-IV, 14%) and 42% (extensive, 24%), respectively. Age ≥ 60 years and Karnofsky Performance Status < 90% at transplant, graft failure, grades III-IV acute GVHD, and disease progression/relapse during follow-up were independently associated with increased mortality24.
Another large retrospective study by the EBMT involving 2,224 patients with myelofibrosis compared myeloablative conditioning (MAC) (35%) vs reduced-intensity conditioning (RIC) (65%) conditioning. There was no significant difference in engraftment, GVHD rates, non-relapse mortality however there was a trend toward a higher relapse rate with RIC regimens compared with MAC (p=0.08). GVHD-free/relapse-free survival (GRFS) at 5 years was 32.4% in the MAC group and 26.1% in the RIC group (p=.001), respectively. The study concluded that MAC should still be used for fit younger individuals suitable in view of trend towards less relapse and an overall advantage of improved GRFS in patients receiving MAC regimens25. In our series, the 1-year mortality rates are high mainly due to sepsis and GVHD with an overall survival of 47%. We need to consider newer strategies to reduce this early mortality by reducing GVHD and sepsis.
A small study from Turkey involving 26 patients showed a 3-year OS rate of 46.2% with lower OS in mismatched unrelated graft recipients compared to MSD and MUD recipients. A study from northern India involving 15 patients with myelofibrosis undergoing allo-SCT showed a OS and DFS were 60% with no relapses at a median follow-up of 364 days. The incidence of acute and chronic GVHD was 27% each and non-relapse mortality was 40%, with the main cause of death being sepsis, followed by acute GVHD21. Table 3 shows comparative results of some studies with our study.
Overall, our results are consistent with other previous studies, underscoring the unmet need regarding allo-SCT in myelofibrosis.
The high proportion of patients who were transfusion-dependent as well as high number of patients with massive splenomegaly at the time of transplant reflects the advanced disease burden in our cohort, which may have influenced post-transplant outcomes. The 2 years OS in our study of 47% highlights the need for more research in this area to improve outcomes. The 2 most important causes of death were GVHD and sepsis. MSD transplants perform better than MUD or haploidentical transplants. MAC is justified in young fit donors than RIC.
There are some limitations of the study. Its retrospective and single-center design with small sample size limits the generalizability of the findings. A uniform protocol for pre-transplant ruxolitinib management was not followed, as the cohort spans both the pre-ruxolitinib and post-ruxolitinib eras, and physician discretion guided its use. Continuation of JAK inhibitor therapy through engraftment is now considered standard practice in many centers, as it may reduce bone marrow niche inflammation and improve engraftment outcomes26. The higher infused stem cell doses used in our cohort, while intended to facilitate engraftment in the setting of splenomegaly and fibrosis, may have contributed to increased GVHD rates. Survival appeared higher in patients transplanted after 2010 compared to those transplanted before 2010, although the cohort size was too small for statistical analysis. Nonetheless it is reasonable to assume that advances in supportive care, antifungal prophylaxis, and GVHD management in the last decade may have contributed to improved outcomes, as reported by larger international series. Despite these limitations, our study contributes valuable data on myelofibrosis transplant outcomes from a low- and middle-income country perspective, highlighting both challenges and opportunities for improvement.
Conclusion
This study, the largest from India, shows reasonable cure rates with allo-SCT for myelofibrosis. We need to evolve newer strategies to reduce GVHD, sepsis associated mortality and graft failure to improve survival in myelofibrosis.
Author Contributions
NJ: Performed research, designed study, analyzed data and wrote the paper, UK: Performed research and analyzed data, SS: Performed research and analyzed data, SL: Performed research and analyzed data, FNA : Performed research and analyzed data, KML: Statistical analysis of data, MM: Performed research and analyzed data, ES : Performed research and analyzed data, PB : Performed research and analyzed data, AK: Performed research and analyzed data, AA: Performed research and analyzed data, VM: Performed research and analyzed data, BG: Performed research, designed study, clinical data accrual, analyzed data, and wrote the paper.
Conflicts of Interest
The authors declare no conflict of interest. Disclosure forms provided by the authors are available on the website.
References
1.Tefferi A, Guglielmelli P, Larson DR, Finke C, Wassie EA, Pieri L, et al. Long-term survival and blast transformation in molecularly annotated essential thrombocythemia, polycythemia vera, and myelofibrosis. Blood. 2014; 124: 2507-13.
2.Jain T, Kunze KL, Mountjoy L, Partain DK, Kosiorek H, Khera N, et al. Early post-transplantation factors predict survival outcomes in patients undergoing allogeneic hematopoietic cell transplantation for myelofibrosis. Blood Cancer J. 2020; 10: 38.
3.Bewersdorf JP, Sheth AH, Vetsa S, Grimshaw A, Giri S, Podoltsev NA, et al. Outcomes of Allogeneic Hematopoietic Cell Transplantation in Patients With Myelofibrosis-A Systematic Review and Meta-Analysis. Transplant Cell Ther. 2021; 27: 873.e1-13.
4.Scott BL, Gooley TA, Sorror ML, Rezvani AR, Linenberger ML, Grim J, et al. The Dynamic International Prognostic Scoring System for myelofibrosis predicts outcomes after hematopoietic cell transplantation. Blood. 2012; 119: 2657-64.
5.Cipkar C, Kumar S, Thavorn K, Kekre N. Optimal Timing of Allogeneic Stem Cell Transplantation for Primary Myelofibrosis. Transplant Cell Ther. 2022; 28: 189-94.
6.Tefferi A. Primary myelofibrosis: 2021 update on diagnosis, risk-stratification and management. Am J Hematol. 2021; 96: 145-62.
7.McLornan DP, Yakoub-Agha I, Robin M, Chalandon Y, Harrison CN, Kroger N. State-of-the-art review: allogeneic stem cell transplantation for myelofibrosis in 2019. Haematologica. 2019; 104: 659-68.
8.Perram J, Ross DM, McLornan D, Gowin K, Kröger N, Gupta V, et al. Innovative strategies to improve hematopoietic stem cell transplant outcomes in myelofibrosis. Am J Hematol. 2022; 97: 1464-77.
9.Teltschik HM, Heinzelmann F, Gruhn B, Feuchtinger T, Schlegel P, Schumm M, et al. Treatment of graft failure with TNI-based reconditioning and haploidentical stem cells in paediatric patients. Br J Haematol. 2016; 175: 115-22.
10.Wolff SN. Second hematopoietic stem cell transplantation for the treatment of graft failure, graft rejection or relapse after allogeneic transplantation. Bone Marrow Transplant. 2002; 29: 545-52.
11.Glucksberg H, Storb R, Fefer A, Buckner CD, Neiman PE, Clift RA, et al. Clinical manifestations of graft-versus-host disease in human recipients of marrow from HL-A-matched sibling donors. Transplantation. 1974; 18: 295-304.
12.Lee SJ, Vogelsang G, Flowers MED. Chronic graft-versus-host disease. Biol Blood Marrow Transplant. 2003; 9: 215-33.
13.Tefferi A, Partain DK, Palmer JM, Slack JL, Roy V, Hogan WJ, et al. Allogeneic hematopoietic stem cell transplant overcomes the adverse survival effect of very high risk and unfavorable karyotype in myelofibrosis. Am J Hematol. 2018; 93: 649-54.
14.Tefferi A, Guglielmelli P, Nicolosi M, Mannelli F, Mudireddy M, Bartalucci N, et al. GIPSS: genetically inspired prognostic scoring system for primary myelofibrosis. Leukemia. 2018; 32: 1631-42.
15.Guglielmelli P, Lasho TL, Rotunno G, Mudireddy M, Mannarelli C, Nicolosi M, et al. MIPSS70: Mutation-Enhanced International Prognostic Score System for Transplantation-Age Patients With Primary Myelofibrosis. J Clin Oncol. 2018; 36: 310-8.
16.Tefferi A, Guglielmelli P, Lasho TL, Gangat N, Ketterling RP, Pardanani A, et al. MIPSS70+ Version 2.0: Mutation and Karyotype-Enhanced International Prognostic Scoring System for Primary Myelofibrosis. J Clin Oncol. 2018; 36: 1769-70.
17.Tefferi A. Primary myelofibrosis: 2023 update on diagnosis, risk-stratification, and management. Am J Hematol. 2023; 98: 801-21.
18.Ballen KK, Shrestha S, Sobocinski KA, Zhang MJ, Bashey A, Bolwell BJ, et al. Outcome of transplantation for myelofibrosis. Biol Blood Marrow Transplant. 2010; 16: 358-67.
19.McLornan D, Szydlo R, Koster L, Chalandon Y, Robin M, Wolschke C, et al. Myeloablative and Reduced-Intensity Conditioned Allogeneic Hematopoietic Stem Cell Transplantation in Myelofibrosis: A Retrospective Study by the Chronic Malignancies Working Party of the European Society for Blood and Marrow Transplantation. Biol Blood Marrow Transplant. 2019; 25: 2167-71.
20.Cengiz Seval G, Civriz Bozdag S, Kocak Toprak S, Kurt Yuksel M, Topcuoglu P, Arslan O, et al. Allogeneic Hematopoietic Stem Cell Transplantation for Primary Myelofibrosis: A 20-year Experience in a Single Center. Balk Med J. 2023; 40: 197-204.
21.Choudhary D, Doval D, Khandelwal V, Setia R, Handoo A. Allogeneic stem cell transplant for myelofibrosis- A retrospective single-center study. Blood Cell Ther. 2023; 6: 5-10.
22.Polverelli N, Mauff K, Kröger N, Robin M, Beelen D, Beauvais D, et al. Impact of spleen size and splenectomy on outcomes of allogeneic hematopoietic cell transplantation for myelofibrosis: A retrospective analysis by the chronic malignancies working party on behalf of European society for blood and marrow transplantation (EBMT). Am J Hematol. 2021; 96: 69-79.
23.Gupta V, Kosiorek HE, Mead A, Klisovic RB, Galvin JP, Berenzon D, et al. Ruxolitinib Therapy Followed by Reduced-Intensity Conditioning for Hematopoietic Cell Transplantation for Myelofibrosis: Myeloproliferative Disorders Research Consortium 114 Study. Biol Blood Marrow Transplant. 2019; 25: 256-64.
24.Hernández-Boluda JC, Pereira A, Kröger N, Beelen D, Robin M, Bornhäuser M, et al. Determinants of survival in myelofibrosis patients undergoing allogeneic hematopoietic cell transplantation. Leukemia. 2021; 35: 215-24.
25.McLornan D, Szydlo R, Koster L, Chalandon Y, Robin M, Wolschke C, et al. Myeloablative and Reduced-Intensity Conditioned Allogeneic Hematopoietic Stem Cell Transplantation in Myelofibrosis: A Retrospective Study by the Chronic Malignancies Working Party of the European Society for Blood and Marrow Transplantation. Biol Blood Marrow Transplant. 2019; 25: 2167-71.
26.Ibrahim U, Petrone GEM, Mascarenhas J, Keyzner A. Peritransplantation Use of Ruxolitinib in Myelofibrosis. Biol Blood Marrow Transplant. 2020; 26: 2177-80.
Search
Actions
News